Designing LPS-Free Protocols for Peptide Reconstitution
Author: Dr. Numan S. Date: March 5, 2026
What Is Peptide Reconstitution?
peptide reconstitution is dissolving a dry (often lyophilized) peptide into a defined solvent to create a measurable stock for experiments. In research workflows, this “first liquid step” sets concentration accuracy and can also set the baseline for endotoxin contamination if solvents or containers carry residual LPS [1].
Because many peptides are used at low nanomolar to micromolar levels, small upstream impurities can disproportionately affect response readouts. Practical peptide preparation methods therefore start by pairing solubility needs with contamination control, treating sterile reconstitution as a microbiology control—distinct from endotoxin control [3,7].
Understanding LPS and Endotoxin Risk
Endotoxin contamination arises primarily from lipopolysaccharides released from Gram-negative bacterial cell membranes. The biologically active portion of LPS, known as lipid A, can trigger strong inflammatory signaling through Toll-like receptor pathways even at extremely low concentrations [2]. Because of this potency, endotoxin contamination can distort experimental results in immunology, cell culture, and pharmacological studies.
Unlike microbial contamination, endotoxins are not living organisms. They can remain active after sterilization processes and may pass through standard membrane filters used for sterile reconstitution [3]. Consequently, laboratory best practices require separate strategies for sterility control and endotoxin removal when designing reliable peptide preparation methods.
Common Sources of LPS During Peptide Reconstitution
Multiple laboratory sources can introduce endotoxin contamination during peptide reconstitution. Water quality is one of the most significant factors because endotoxins can accumulate in purification systems or laboratory storage containers. Even highly purified water may contain trace LPS if storage conditions allow microbial growth or biofilm formation [4].

Figure 1: Common contamination sources and solutions
Other common contamination pathways include laboratory air exposure, reusable glassware, plastic consumables, and improper peptide handling procedures. Research has shown that many commercial reagents used in biological research may carry measurable endotoxin levels if not specifically tested or treated [1]. As a result, LPS-free protocols typically require endotoxin-tested water, depyrogenated glassware, and controlled laboratory workflows for research peptide handling.
Designing LPS-Free Reconstitution Protocols
Well-designed LPS-free protocols treat peptide reconstitution as a controlled process rather than a simple mixing step. Researchers typically begin by selecting endotoxin-free water sources such as water for injection (WFI) or certified ultrapure laboratory water. The use of sterile reconstitution containers and single-use pipette tips further reduces contamination risk.
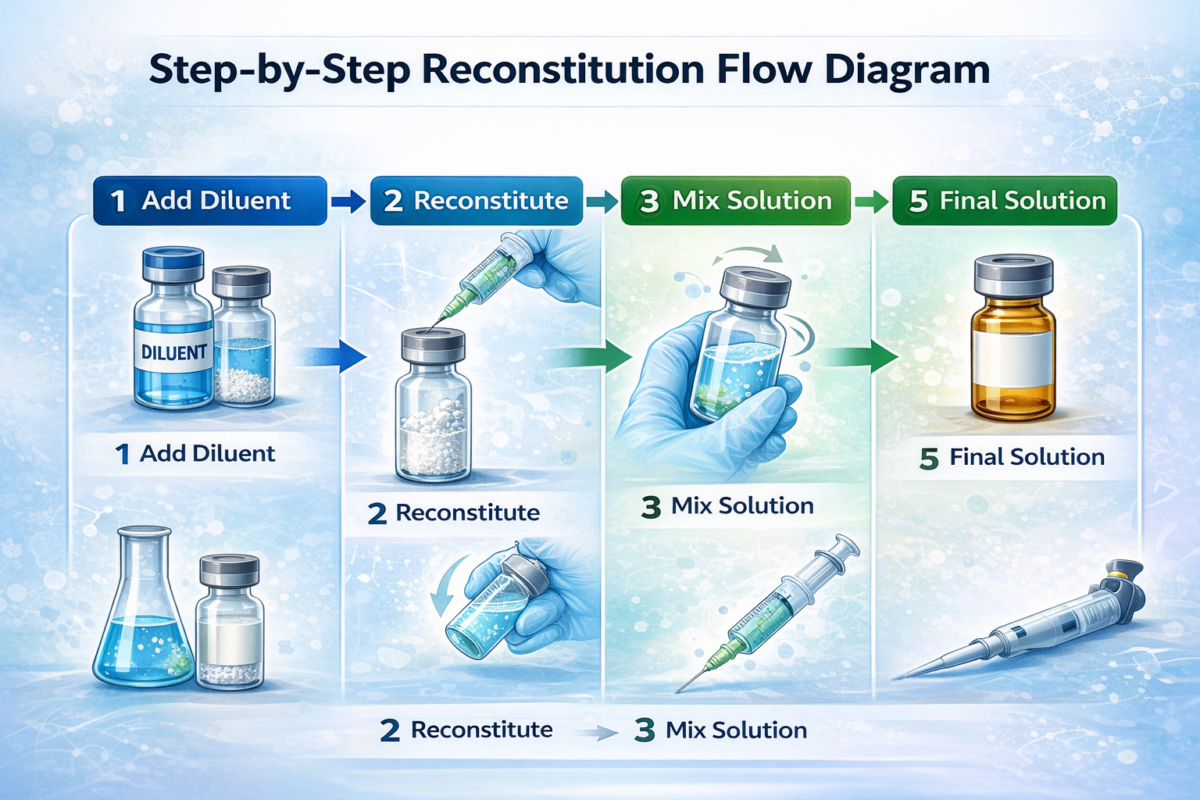
Figure 2: Step-by-step reconstitution guide
Another important strategy involves depyrogenation procedures. Dry heat treatment at temperatures around 250 °C has been shown to significantly reduce endotoxin levels on compatible glass materials, providing an effective endotoxin removal approach when preparing containers and equipment [5]. Incorporating these controls into peptide handling procedures helps establish standardized peptide preparation methods that reduce experimental variability.
Detecting and Monitoring Endotoxin After Reconstitution
Endotoxin testing is typically performed after peptide reconstitution to verify that contamination levels remain below acceptable thresholds. The most widely used assay is the Limulus Amebocyte Lysate (LAL) test, which detects LPS through an enzymatic clotting cascade derived from horseshoe crab blood [7].
Alternative assays, including recombinant Factor C (rFC) tests, also measure endotoxin activity without animal-derived reagents. Many laboratories perform testing immediately after peptide reconstitution and again after major process changes, such as new solvents or equipment. Monitoring endotoxin contamination at these checkpoints helps ensure that peptide handling procedures remain reliable.
Best Practices for Handling and Storage Post-Reconstitution
After peptide reconstitution, proper storage conditions are critical for maintaining sample integrity. Researchers often divide reconstituted solutions into small aliquots to avoid repeated vial entry. This practice reduces the likelihood that endotoxin contamination will be introduced during repeated pipetting or sampling events.
Cold storage also supports contamination control. Many peptide preparation methods recommend storing aliquots at −20 °C or colder to minimize microbial growth and preserve peptide stability. These steps are part of broader laboratory best practices for sterile reconstitution and research peptide handling workflows designed to maintain solution purity over time.
Reconstitution Protocols Shape Endotoxin Risk
Ultimately, peptide reconstitution protocols strongly influence the likelihood of endotoxin contamination. Assumptions that sterile filtration alone prevents contamination can lead to misleading results, since endotoxins are small enough to pass through typical sterilizing filters [3]. Instead, laboratories must combine sterile reconstitution with dedicated endotoxin removal strategies.
Building reproducible protocols involves defining approved solvents, validated equipment preparation methods, and consistent peptide handling procedures. When laboratories implement structured LPS-free protocols supported by endotoxin monitoring, peptide preparation methods become more reproducible and experimental outcomes become more reliable. In research peptide handling environments, careful protocol design is therefore essential for minimizing hidden endotoxin risks.
Frequently asked questions (FAQs) about Endotoxin Risk During Peptide Reconstitution
Why is peptide reconstitution a major endotoxin risk point?
- Peptide reconstitution is often considered a critical endotoxin risk point because it is the stage where a previously lyophilized peptide is exposed to water, laboratory surfaces, and handling tools. Lipopolysaccharide (LPS), the endotoxin component of Gram-negative bacterial outer membranes, is highly stable and can persist on glassware, pipettes, and laboratory water systems. Even trace contamination introduced during reconstitution can remain associated with the peptide solution because endotoxins resist heat, filtration, and many sterilization approaches [1,2]. For this reason, laboratories performing peptide research often treat the reconstitution step as a controlled procedure with careful water selection, sterile technique, and contamination monitoring.
What protocol steps most influence LPS contamination?
- Several protocol variables strongly influence the likelihood of endotoxin introduction during peptide preparation. Water quality is one of the most important factors; ultrapure laboratory water or endotoxin-tested water systems are commonly used to reduce contamination risk. Container choice also matters because endotoxins can adsorb to surfaces and later release into solution. Additionally, repeated opening of containers, non-sterile pipette tips, and environmental exposure increase the chance that environmental bacterial residues will introduce LPS into the preparation workflow [3]. Proper handling procedures therefore emphasize minimizing exposure time and using endotoxin-controlled materials whenever possible.
How can researchers reduce endotoxin during reconstitution?
- Researchers commonly reduce endotoxin exposure by implementing laboratory protocols designed to limit contamination. These include preparing solutions in controlled work areas, using certified endotoxin-free consumables, and selecting high-purity water sources such as Type I ultrapure water or water for injection–grade laboratory systems when appropriate for research workflows. Some laboratories also perform routine environmental monitoring and endotoxin testing of water systems or reagents to verify that contamination levels remain low [4]. When consistent protocols are followed, the probability of introducing LPS during reconstitution can be significantly reduced.
What peptide properties increase LPS binding or masking?
- Certain peptide characteristics can influence how endotoxin behaves in solution. Positively charged peptides, amphiphilic structures, or sequences with hydrophobic regions may interact with the lipid A portion of LPS through electrostatic or hydrophobic interactions. These interactions can promote binding between peptides and endotoxin molecules, which in some cases can alter assay detection or distribution within a sample matrix [5]. Because of these potential interactions, researchers sometimes evaluate assay interference or endotoxin masking when testing peptide preparations.
How should labs document low-endotoxin preparation workflows?
- Laboratories typically document low-endotoxin workflows through written standard operating procedures (SOPs) that define acceptable materials, water specifications, environmental controls, and handling steps. Documentation may include batch preparation records, water quality verification logs, endotoxin test results, and equipment cleaning protocols. Maintaining clear documentation helps ensure reproducibility and provides traceability for research records, particularly when peptide samples are analyzed or shared across different laboratories [1,4]. Well-documented workflows are an important part of quality-focused peptide research practices.
References
- Weinstein JR, Swarts S, Bishop C, Hanisch UK, Möller T. Lipopolysaccharide is a frequent and significant contaminant in microglia-activating factors. Glia. 2008;56(1):16-26.
- Heinbockel L, Weindl G, Martinez-de-Tejada G, et al. Inhibition of lipopolysaccharide- and lipoprotein-induced inflammation by antitoxin peptide Pep19-2.5. Front Immunol. 2018;9:1704.
- Bononi I, Puggioli G, Faccini R, et al. Gram-negative bacterial lipopolysaccharide retention by a positively charged new-generation filter. Appl Environ Microbiol. 2008;74(21):6841-6849.
- Li Y, Boraschi D. Endotoxin contamination in nanomaterials leads to the misinterpretation of immunosafety results. Front Immunol. 2017;8:472.
- Li L, Tsai JJ, Li P. Kinetics of hydrothermal inactivation of endotoxins. Appl Environ Microbiol. 2011;77(23):8598-8607.
- U.S. Food and Drug Administration. Guidance for Industry: Pyrogen and Endotoxins Testing: Questions and Answers. June 2012. Accessed March 8, 2026.
- U.S. Food and Drug Administration. High purity water system (7/93). Updated 2016. Accessed March 8, 2026.
- Kaconis Y, Kowalski I, Howe J, et al. Biophysical mechanisms of endotoxin neutralization by cationic amphiphilic peptides. Biophys J. 2011;100(11):2652-2661.
