Glutathione Peptide (1500MG)
This batch of L-Glutathione has been third party lab tested and verified for quality.
TESTED FOR:

purity

weight

endotoxins(lps)
$79.99
| Discount | Quantity | Price |
|---|---|---|
| 0% | 1 - 4 | $79.99 |
| 10% Off | 5 - 9 | $71.99 |
| 15% Off | 10 - 24 | $67.99 |
| 25% Off | 25 + | $59.99 |

SHIPS TODAY
Order by 1:00 PM EST

FREE SHIPPING
For orders over $200

ICE PACK SHIPPING
Styrofoam box shipping available
Bottle: lyophilized vial – sealed – flip top
Vial size: large 20ML
Form: powder (lyophilized)
Not reconstituted
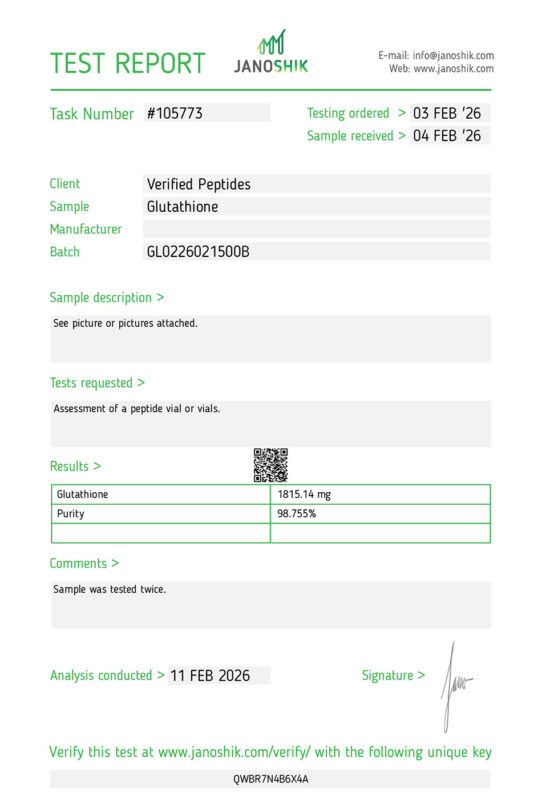
| Glutathione Test Results | Results | |
|---|---|---|
Glutathione Test results | ||
Date Tested: | February 11, 2026 | |
Purity: | 98.755% | |
Weight: | 1815.14MG | |
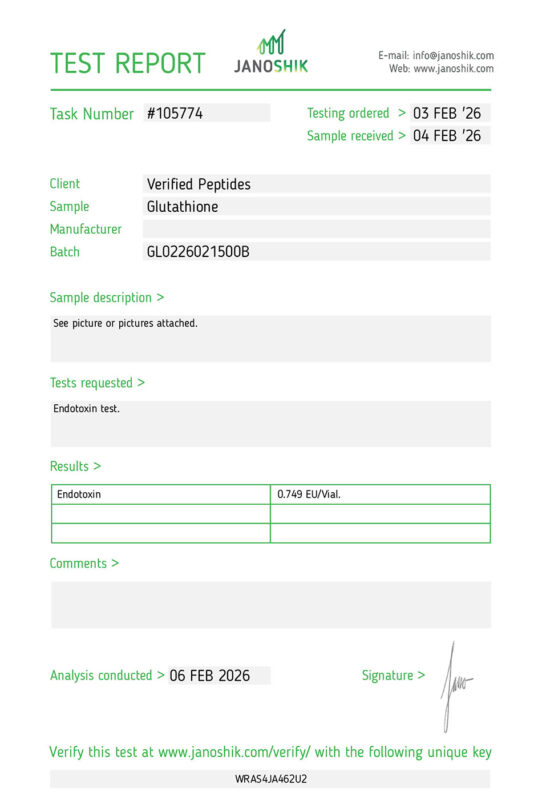
Endotoxins (LPS): | Pass | |
Batch #: | GL0226021500B | |

Glutathione Peptide Information
FORM |
Powder (lyophilized) |
|---|---|
CAS NUMBER |
70-18-8 |
SEQUENCE |
C10H17N3O6S |
OTHER NAMES |
reduced glutathione, glutathion |
WEIGHT |
1500mg |
Molecular Weight |
307.32 g/mol |
Terms |
Subject to our Terms and Conditions. This material is sold for laboratory research use only. Not for human consumption, animal, or medical use. |
Why isn’t there more information on Glutathione peptide?
Due to the legal landscape of peptides and research products, providing information that may imply anything beyond laboratory research use is a legal liability. We’re an expert biotechnology company that provides high quality peptides and products for purchase to advance scientific research in this field.

Why Choose Verified Peptides?
The industry's most trusted source for research peptides
The #1 Leader In Peptide Testing
400+ Certificates of Analysis
What does this mean for you? You can be sure we don't cut corners on testing every single batch.
Visit our Lab Reports page.
A Long Standing Trusted Company
6+ Years of Peptides
We pioneered third party lab testing and have stood the test of time. Our first lab report dates back to 2019 proving our track record.
The Best $/mg Prices At The Highest Quality
You won't find this degree of testing and quality at this price.
Peptides Tested For More Than Just Purity
We conduct an array of tests via HPLC and other methods for our peptides:
purity, weight, endotoxins(LPS), sterility (bacteria & mold/yeast), and TFA content.
Transparency. Trusted. Reliable.
- Batch number on every vial
- Published Certificates of Analysis
- Expert customer service
- Satisfaction and quality guarantee
- Cold pack styrofoam shipping
- Secure credit card payment
Explore Other High-Purity Peptides
Hear What Our Customers Say
Trusted by thousands of researchers for high-quality peptides
Testing Antioxidants Against Age-Related Oxidative Stress: What a Rat Model Tells Us About the Glutathione Antioxidant Peptide
This article summarizes a controlled animal study examining whether a combination of vitamin C and glutathione — a naturally occurring glutathione antioxidant tripeptide — could reduce oxidative stress markers and modulate related biochemical changes in aging male rats.
Background: Why Glutathione Is Called an Antioxidant "Peptide"
Glutathione (GSH) is classified as a tripeptide — a short-chain molecule composed of three amino acids — that serves a central role in the body's cellular antioxidant defense systems. As a glutathione peptide, it functions not as a simple free-radical scavenger alone, but as an essential substrate for two major antioxidant enzymes: glutathione peroxidase and glutathione-S-transferase. These enzymes depend on GSH to neutralize reactive oxygen species (ROS) and detoxify harmful compounds within cells. The peptide's biochemical identity — small, water-soluble, and enzymatically active — is precisely why it occupies a prominent position in oxidative stress research.
Vitamin C (ascorbic acid, or AsA) was paired with glutathione in the intervention studied here because of the proposed interaction between the two molecules. The study's authors cite prior research suggesting that ascorbic acid may participate in the direct reduction of disulfide bonds and in supporting the glutathione reductase mechanism responsible for regenerating reduced GSH from its oxidized form (GSSG). In this way, vitamin C may act as a cofactor that helps maintain and recycle the glutathione antioxidant pool within tissues — an important consideration given that GSH can be depleted under conditions of chronic oxidative stress, such as aging.
GSH is synthesized intracellularly from glutamate, cysteine, and glycine. Its tripeptide structure is what earns it the designation as a glutathione peptide — a designation that distinguishes it from larger proteins and positions it within a class of bioactive short-chain molecules with measurable physiological functions.
Study Objective and Why This Research Matters
The central objective of this study was to determine whether supplementing older male rats with a combined daily dose of vitamin C and GSH could reduce age-associated oxidative stress and modulate downstream biochemical changes. Aging is widely recognized as a condition associated with the accumulation of free radical-induced cellular damage. The study authors frame the biological rationale clearly: oxygen-based free radicals accumulate over time and contribute to age-related tissue deterioration, making antioxidant supplementation a potentially meaningful intervention in older animals.
The practical importance of the study lies in the breadth of outcomes measured. Rather than examining oxidative damage in isolation, the researchers tracked a comprehensive panel of readouts: tissue lipid peroxidation, tissue glutathione status, serum lipid profiles, hormonal markers (testosterone, T3, and T4), and trace mineral concentrations (copper and zinc) across multiple compartments. This multi-endpoint design allows readers to assess whether vitamin C and glutathione antioxidant supplementation produced changes that extended beyond direct oxidative stress markers into broader metabolic and endocrine physiology — an important consideration in evaluating antioxidant interventions in the aging context.
How the Animal Study Was Designed
The study used 18-month-old male Sprague–Dawley rats, selected as an established aging model. At this age, these animals exhibit documented increases in oxidative stress and age-related biochemical shifts comparable to a geriatric physiological state. Rats weighed between 500 and 550 grams at the time of the experiment, indicating a healthy cohort of older males.
Animals were divided into two groups of five each: (a) a control group of untreated aged rats, and (b) a supplemented group that received a daily intragastric administration of the antioxidant mixture for six consecutive weeks. The supplement consisted of vitamin C at 30 mg/kg body weight combined with GSH at 100 mg/kg body weight — a formulation selected based on prior published work cited by the author. At the conclusion of the six-week period, all animals were sacrificed, and tissue and blood samples were collected for analysis.
Oxidative stress: TBARS (thiobarbituric acid-reactive substances) in liver and testes — a standard proxy for lipid peroxidation.
Tissue antioxidant status: GSH concentration in liver and testicular tissue.
Hormones: Serum testosterone, triiodothyronine (T3), and thyroxine (T4).
Serum lipids: Total cholesterol and triglycerides.
Trace minerals: Copper (Cu) and zinc (Zn) in serum, liver, brain, and testes.
Key Findings: Lower Oxidative Stress Markers in Aging Rats
TBARS Decreased in Liver and Testes
The most directly relevant outcome for assessing the glutathione antioxidant intervention was the measurement of TBARS — thiobarbituric acid-reactive substances — as a marker of lipid peroxidation (LPO). Supplemented aged rats showed a statistically significant reduction in TBARS concentrations in both the liver and the testes compared with non-supplemented aged controls. Hepatic TBARS values in control animals averaged approximately 3,933 nmol/g fresh tissue versus 2,546 nmol/g in supplemented animals. In testicular tissue, the difference was more pronounced: control animals averaged roughly 3,160 nmol/g compared to approximately 1,164 nmol/g in the supplemented group. Both reductions were significant at p < 0.05, consistent with meaningfully lower lipid peroxidation — and therefore reduced oxidative tissue damage — in the animals receiving vitamin C plus GSH.
Tissue GSH Changed Selectively
The pattern of tissue glutathione peptide response was notably organ-specific. Testicular GSH levels increased significantly after the six-week supplementation period, rising from a mean of approximately 0.65 µmol/g fresh tissue in control animals to approximately 1.95 µmol/g in supplemented rats (p < 0.05). Hepatic GSH, by contrast, did not show a statistically meaningful change — values of roughly 1.83 µmol/g in controls and 1.84 µmol/g in the treated group were virtually identical. This tissue-specific response is an important observation: the study demonstrates that the glutathione antioxidant effects of this combined supplementation were not uniform across organs, and that the testes appeared to be the primary site of GSH augmentation under these conditions.
| Parameter | Tissue | Control (Mean ± SE) | Supplemented (Mean ± SE) | Significance |
|---|---|---|---|---|
| TBARS (nmol/g) | Liver | 3,933 ± 298.8 | 2,546 ± 148.3 | p < 0.05 ↓ |
| TBARS (nmol/g) | Testes | 3,160 ± 318.8 | 1,164 ± 70.49 | p < 0.05 ↓ |
| GSH (µmol/g) | Liver | 1.83 ± 0.01 | 1.84 ± 0.08 | Not significant |
| GSH (µmol/g) | Testes | 0.65 ± 0.02 | 1.95 ± 0.09 | p < 0.05 ↑ |
Metabolic and Hormonal Readouts
Serum Lipids Improved in the Supplemented Group
Both serum cholesterol and triglyceride levels were significantly lower in animals receiving the vitamin C plus glutathione antioxidant combination. Serum cholesterol fell from a mean of approximately 84.4 mg/dL in controls to 67.4 mg/dL in supplemented rats (p < 0.05). Triglycerides similarly declined, from roughly 105.68 mg/dL in controls to 86.26 mg/dL in the supplemented group (p < 0.05). The authors interpret this alongside the reduction in TBARS as evidence that the ameliorated oxidative state may have downstream consequences for lipid metabolism in older animals.
Testosterone Increased, Thyroid Hormones Did Not
Serum testosterone levels were significantly higher in supplemented aged rats compared with controls, rising from approximately 75.8 ng/dL to 113.68 ng/dL (p < 0.05). The study authors attribute this increase to the reduction of oxidative stress in testicular tissue and the concomitant rise in testicular GSH — reflecting the known sensitivity of steroidogenic function in the testes to oxidative damage. Both GSH and vitamin C, the authors note, may play a role in regulating the membrane redox state of endocrine secretory tissue, which could in turn influence testosterone output.
In contrast, thyroid hormones were not significantly affected. Serum T3 values were 0.9 ± 0.07 ng/dL in controls and 0.92 ± 0.06 ng/dL in supplemented animals — a negligible difference. T4 values similarly showed no meaningful change (5.0 ± 0.33 vs. 4.52 ± 0.48 ng/dL). The study's authors conclude that vitamin C plus glutathione peptide supplementation at these doses and durations did not influence thyroid activity in aging rats, which aligns with prior findings cited in the paper regarding the lack of a significant correlation between antioxidant enzyme activity and T3/T4 levels in young and old rats.
| Serum Parameter | Control | Supplemented | Significance |
|---|---|---|---|
| Testosterone (ng/dL) | 75.8 ± 4.3 | 113.68 ± 6.2 | p < 0.05 ↑ |
| T3 (ng/dL) | 0.9 ± 0.07 | 0.92 ± 0.06 | Not significant |
| T4 (ng/dL) | 5.0 ± 0.33 | 4.52 ± 0.48 | Not significant |
| Cholesterol (mg/dL) | 84.4 ± 2.9 | 67.4 ± 1.8 | p < 0.05 ↓ |
| Triglycerides (mg/dL) | 105.68 ± 4.34 | 86.26 ± 4.6 | p < 0.05 ↓ |
Trace Minerals: Copper and Zinc Shifts
A notable set of findings from this study involves the distribution of the trace minerals copper (Cu) and zinc (Zn) across serum and tissues. In supplemented aged rats, copper concentrations were significantly elevated in all four measured compartments: serum (1.19 vs. 1.84 µg/mL), liver (2.136 vs. 2.487 µg/g), brain (2.137 vs. 3.741 µg/g), and testes (1.931 vs. 2.737 µg/g) — all at p < 0.05. The most pronounced Cu elevation was observed in brain tissue.
Zinc concentrations increased significantly in serum (2.31 vs. 4.59 µg/mL) and brain (14.93 vs. 20.591 µg/g), but did not show statistically significant changes in liver or testes Zn concentrations. The authors note that both copper and zinc exert their biological roles as catalysts in association with specific proteins — including enzymes involved in antioxidant defense — which may explain why their tissue distributions shifted in response to altered oxidative state and glutathione antioxidant activity. However, the exact mechanistic pathway between antioxidant supplementation and these trace mineral changes is not elaborated upon in this particular study.
| Mineral | Compartment | Control | Supplemented | Significance |
|---|---|---|---|---|
| Zn (µg/mL) | Serum | 2.31 ± 0.19 | 4.59 ± 0.57 | p < 0.05 ↑ |
| Zn (µg/g) | Liver | 14.953 ± 1.38 | 11.983 ± 0.73 | Not significant |
| Zn (µg/g) | Brain | 14.93 ± 0.662 | 20.591 ± 0.59 | p < 0.05 ↑ |
| Cu (µg/mL) | Serum | 1.19 ± 0.24 | 1.84 ± 0.14 | p < 0.05 ↑ |
| Cu (µg/g) | Liver | 2.136 ± 0.05 | 2.487 ± 0.057 | p < 0.05 ↑ |
| Cu (µg/g) | Brain | 2.137 ± 0.13 | 3.741 ± 0.29 | p < 0.05 ↑ |
| Cu (µg/g) | Testes | 1.931 ± 0.09 | 2.737 ± 0.22 | p < 0.05 ↑ |
Mechanism Context: Antioxidant "Recycling" and Tissue Protection
A key theoretical framework offered by the study is the concept of antioxidant recycling. The authors propose that the combination of vitamin C and the glutathione peptide may work in concert to stimulate an interlocking set of antioxidant pathways. Ascorbic acid, in this model, is thought to support the glutathione reductase mechanism and activate antioxidant recycling pathways, thereby helping to regenerate reduced glutathione. The restored GSH pool is then available as a substrate for glutathione peroxidase and related enzymes, enabling continued neutralization of lipid peroxidation products — reflected in the study's observed TBARS reductions.
The tissue-specific pattern of results provides an important nuance for interpreting this mechanism. While testicular tissue showed both a significant rise in local GSH and a significant drop in TBARS, hepatic tissue showed only a reduction in TBARS without any meaningful increase in tissue GSH. This dissociation indicates that the liver may have other available antioxidant compensatory mechanisms, or that the supplementation influenced enzymatic activity at that site rather than altering the pool of the free glutathione antioxidant tripeptide directly. These organ-level differences highlight that antioxidant responses are not monolithic and should be interpreted in a tissue-specific context in aging models.
How This Fits Into Broader "Glutathione Peptide" Research
This study represents a foundational preclinical example connecting antioxidant supplementation — specifically the combination of vitamin C with the glutathione antioxidant tripeptide — to a range of measurable physiological endpoints in older animals. Its value within the broader literature on glutathione peptides lies in the way it operationalizes GSH's biochemical identity as a peptide: rather than treating it as a generic antioxidant, the study designs an intervention around GSH's specific role as a substrate in enzymatic recycling pathways and measures outcomes that are mechanistically downstream of that function.
For readers seeking research on glutathione peptides in the context of aging and oxidative stress, this study provides a multi-endpoint snapshot: TBARS as a direct lipid peroxidation marker, tissue GSH as a measure of local antioxidant status, and broader physiological readouts (cholesterol, triglycerides, testosterone, trace minerals) as indicators of systemic biochemical state. It is important to note, however, that all of these findings are restricted to an animal model and cannot be directly extrapolated to human physiology.
Important Clarity on Use and Dosing
All findings reported in this summary come exclusively from a controlled study in 18-month-old male Sprague–Dawley rats receiving daily intragastric doses of vitamin C (30 mg/kg body weight) and glutathione (100 mg/kg body weight) over a six-week period. These dosages and the associated outcomes are specific to this animal model and experimental design. They do not constitute, and should not be interpreted as, guidance for human use.
Conclusion
In older male Sprague–Dawley rats, six weeks of daily intragastric supplementation with vitamin C plus the glutathione antioxidant peptide produced a consistent and statistically significant pattern of change across multiple biochemical domains. TBARS — the study's primary index of lipid peroxidation and oxidative stress — declined significantly in both liver and testicular tissue. Testicular glutathione peptide levels rose substantially, while hepatic GSH remained unchanged, illustrating the organ-specificity of antioxidant responses in this model. Serum cholesterol and triglycerides decreased, testosterone increased, thyroid hormone levels were not significantly affected, and both copper and zinc concentrations shifted in serum and select tissues — with copper rising in all four compartments measured and zinc rising significantly in serum and brain.
Collectively, these results contribute to the preclinical evidence base on the capacity of combined glutathione antioxidant and vitamin C supplementation to modulate age-associated oxidative damage and related biochemical markers in an aging rat model. While findings from this study are restricted to the animal model in which they were generated, they provide a structured example of how researchers have investigated glutathione peptides as part of a broader antioxidant strategy against the biochemical consequences of aging.
At a Glance: What Changed vs. What Didn't
Frequently Asked Questions
https://www.jstor.org/stable/48507271