Comparing LPS Detection Methods: LAL, rFC, and MAT Assays
Author: Dr. Numan S. Date: February 18, 2026

Explore the most common LPS detection methods and how they work, where they differ, and what researchers should consider when selecting an approach.
What Is LPS and Why Detection Matters
Lipopolysaccharide (LPS) is a structural component of the outer membrane of Gram-negative bacteria and a potent trigger of inflammatory immune responses. Even trace contamination can influence cytokine signaling, immune assays, and cell culture experiments. For this reason, LPS detection is an essential quality-control step in pharmaceutical development and laboratory research workflows [1,2].
In research environments, LPS detection typically refers to identifying and quantifying endotoxin activity in biological samples, reagents, or manufactured products. Because LPS can trigger strong immune responses even at extremely low concentrations, reliable endotoxin detection helps prevent experimental artifacts and improves reproducibility across studies [2,3].
Overview of LPS Detection Methods
Several laboratory methods are used for LPS detection, with the most common approaches based on Factor C activation or immune-cell responses. Factor C assays include both the traditional LAL assay and the newer rFC assay, while the MAT assay uses human immune cells to measure cytokine release after exposure to pyrogens [3,4].
These approaches differ in what they detect. The LAL assay and rFC assay are designed primarily for endotoxin detection, meaning they specifically respond to LPS molecules. By contrast, the MAT assay detects immune activation from multiple pyrogen sources, including endotoxin and certain non-endotoxin stimuli [4,5].
Limulus Amebocyte Lysate (LAL) Assays
The LAL assay is historically the most widely used method for LPS detection. It is derived from the blood cells (amebocytes) of the horseshoe crab Limulus polyphemus. When LPS is present, it activates the Factor C enzyme within the lysate, triggering a protease cascade that ultimately produces a measurable gel clot or chromogenic signal [3].
This enzymatic cascade allows highly sensitive endotoxin detection, often capable of detecting extremely low endotoxin levels. However, the LAL assay can be affected by interfering substances such as β-glucans that activate a parallel pathway. To prevent false signals during LPS detection, many laboratories use glucan-blocking reagents or specialized assay formats [1,3].
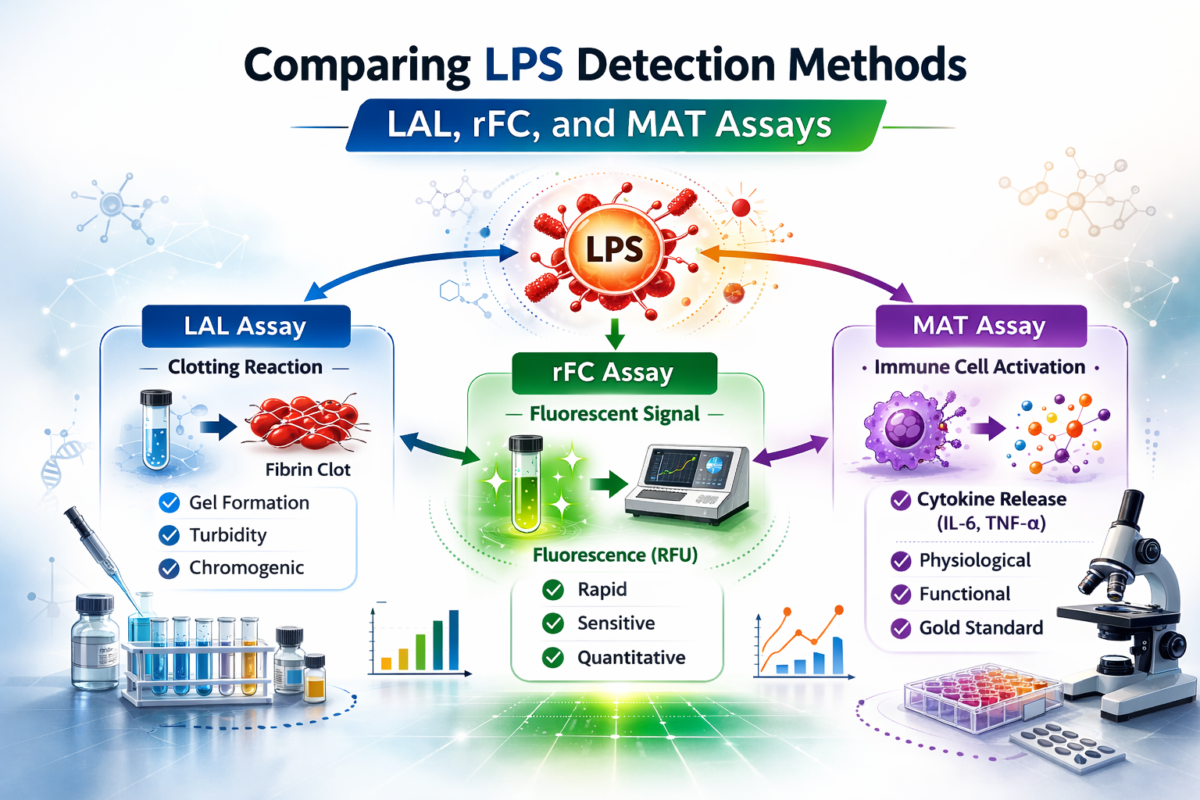
Figure 1: Comparing LPS Detection Methods
Recombinant Factor C (rFC) Assays
The rFC assay was developed as an alternative Factor C–based system that does not rely on horseshoe crab lysate. Instead, it uses recombinant Factor C protein combined with a fluorogenic substrate to detect LPS-driven activation [3,6].
One advantage of the rFC assay is improved specificity for LPS detection, since it lacks the Factor G pathway responsible for β-glucan interference in some LAL systems. Studies comparing the LAL assay and rFC assay have shown strong agreement in many applications, including environmental samples and pharmaceutical testing, although performance can still vary depending on the sample matrix [1,2,6].
Monocyte Activation Test (MAT)
The MAT assay is fundamentally different from Factor C assays because it measures biological responses rather than enzymatic activation. In this method, human monocytes or peripheral blood mononuclear cells are exposed to the sample, and cytokine release—often interleukin-6 or interleukin-1β—is measured as an indicator of pyrogenic activity [4].
Unlike traditional LPS detection methods, the MAT assay can detect both endotoxin and non-endotoxin pyrogens that stimulate immune receptors. Because of this broader sensitivity, the MAT assay is sometimes used to evaluate overall pyrogenic risk rather than strictly endotoxin contamination [4,5].
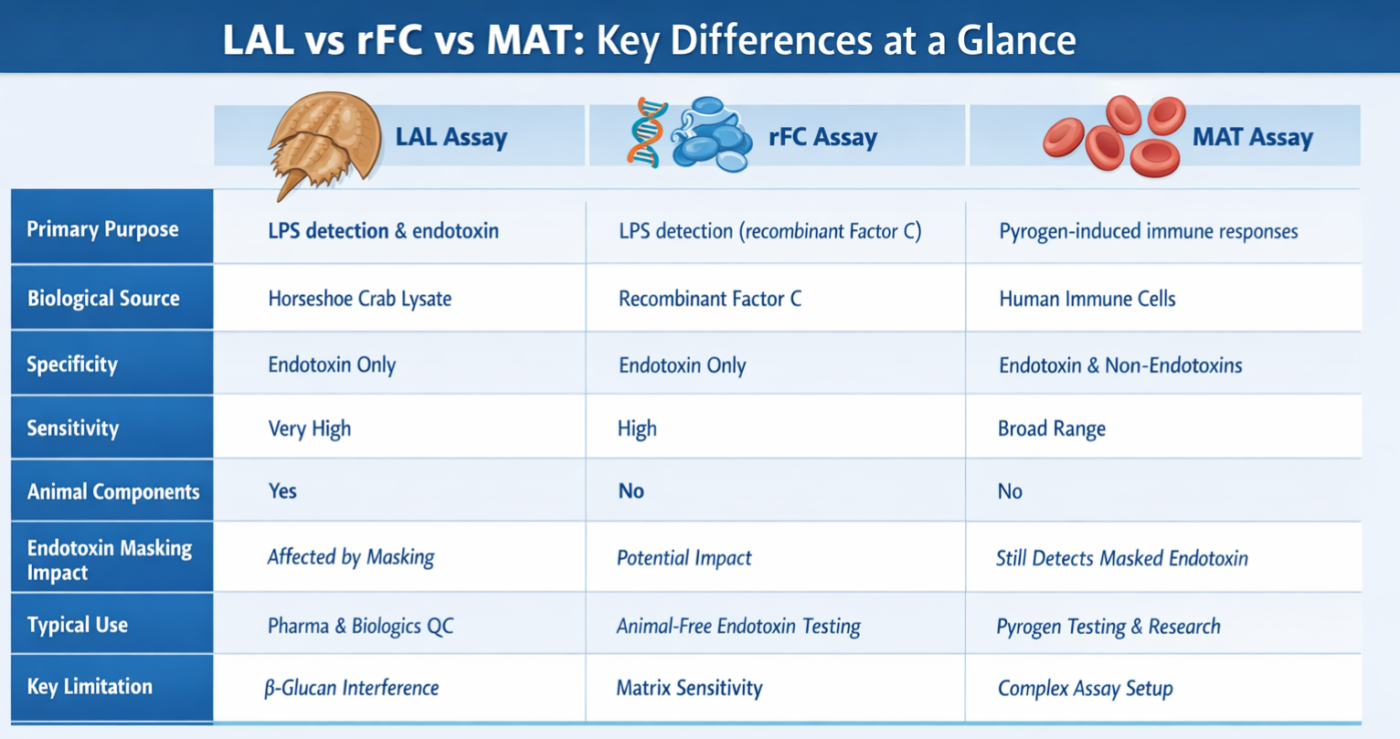
Figure 2: Assay comparison: LAL, rFC, MAT
LAL vs rFC vs MAT: Key Differences at a Glance
The LAL assay and rFC assay share a common biochemical principle: both rely on Factor C activation for endotoxin detection. These assays are often used for rapid screening because they are sensitive, standardized, and compatible with high-throughput laboratory workflows [3].
The MAT assay, in contrast, measures cytokine production from human immune cells. This means it can detect endotoxin as well as other pyrogenic contaminants. As a result, MAT is often considered a broader biological test rather than a strictly analytical LPS detection method [4].
Interference and Masking Challenges in LPS Detection
One of the major analytical challenges in LPS detection is interference from sample components. Certain buffers, surfactants, or biological matrices can alter the accessibility of endotoxin molecules during testing.
A particularly important phenomenon is endotoxin masking, also called low endotoxin recovery. In this situation, endotoxin may be present but becomes difficult to detect due to interactions with detergents, chelators, or formulation components. This can reduce apparent endotoxin detection signals even when biologically active endotoxin remains in the sample [8,9].
Research has shown that endotoxin masking may affect Factor C–based assays such as the LAL assay and rFC assay, potentially leading to underestimation of endotoxin levels. Importantly, masked endotoxin can still stimulate immune responses in cell-based systems such as the MAT assay, demonstrating that endotoxin masking does not necessarily eliminate biological activity [8,10].
Choosing the Right LPS Detection Method for Research Use
Selecting the best LPS detection strategy depends on the research goal and sample composition. If the objective is rapid and sensitive endotoxin detection, Factor C–based assays such as the LAL assay or rFC assay are often preferred because they are well characterized and widely validated [3].
However, when complex formulations or multiple pyrogen sources are possible, researchers may use the MAT assay to capture broader immune activation responses. Understanding the strengths and limitations of each LPS detection method helps laboratories interpret results more accurately and select appropriate quality-control strategies [4].
Why No Single LPS Detection Method Fits Every Use Case
No single LPS detection technique is universally optimal. Each method reflects a different biological or biochemical principle, which means that results can vary depending on endotoxin structure, matrix composition, and assay design.
For this reason, many laboratories use orthogonal testing strategies. A LAL assay or rFC assay may be used for routine endotoxin detection, while the MAT assay can serve as a complementary method when broader pyrogen responses are a concern or when endotoxin masking may influence Factor C results [4,8].
Understanding LPS Detection Methods Supports Better Research Outcomes
Reliable LPS detection is critical for maintaining data integrity in biomedical research and pharmaceutical development. Because endotoxin contamination can influence immune signaling, experimental reproducibility, and product safety, careful assay selection is essential.
By understanding how the LAL assay, rFC assay, and MAT assay differ—and how factors like endotoxin masking may influence results—researchers can design more reliable testing workflows and interpret endotoxin measurements with greater confidence.
Frequently asked questions (FAQs) about LPS Detection Methods
What are the main methods for LPS detection?
- The most widely used laboratory methods for detecting lipopolysaccharide (LPS), commonly referred to as endotoxin, include Limulus Amebocyte Lysate (LAL) assays, recombinant Factor C (rFC) assays, and the Monocyte Activation Test (MAT).
- LAL assays are based on a coagulation cascade found in horseshoe crab blood cells that reacts specifically to endotoxin. Recombinant Factor C assays use a genetically engineered version of the endotoxin-sensitive protein from this same pathway, eliminating the need for animal-derived reagents. The Monocyte Activation Test differs from both methods by measuring human immune cell activation in response to pyrogens, including endotoxin and certain non-endotoxin inflammatory compounds.
- Each method detects endotoxin through a different biological mechanism, which can influence sensitivity, specificity, and how the assay performs with complex samples [1][2].
How do LAL, rFC, and MAT assays differ?
- Although all three methods are used to detect endotoxin or pyrogenic activity, they rely on distinct detection principles.
- LAL assays measure the activation of a natural enzymatic cascade triggered when endotoxin interacts with proteins in horseshoe crab amebocytes. The result can be detected through gel clot formation, turbidity changes, or chromogenic signals. Recombinant Factor C assays isolate the first step of this endotoxin-triggered cascade, using a synthetic form of Factor C that produces a fluorescent signal when activated by LPS.
- The Monocyte Activation Test uses human immune cells or monocyte cell lines that release cytokines when exposed to pyrogens. Because of this mechanism, MAT can detect a broader range of pyrogenic substances beyond endotoxin alone. These differences mean that LAL and rFC are typically used for direct endotoxin quantification, while MAT is often applied when broader pyrogen detection is required [3][4].
Which LPS detection method is most specific?
- Among the commonly used assays, recombinant Factor C (rFC) assays are generally considered the most specific for endotoxin detection.
- This specificity comes from the fact that rFC assays use a single purified recombinant protein—Factor C—that is activated directly by endotoxin’s lipid A component. In contrast, traditional LAL assays contain multiple enzymes from the horseshoe crab coagulation cascade, which can sometimes react with other substances such as β-glucans, potentially producing false positives in certain conditions.
- Because rFC assays isolate the key endotoxin-triggered step, they reduce interference from non-endotoxin compounds and provide a more targeted measurement of LPS [2][5].
How does endotoxin masking affect assay performance?
- Endotoxin masking occurs when components in a sample bind to or alter endotoxin molecules, preventing them from interacting normally with detection reagents. This can cause endotoxin levels to appear artificially low or undetectable during testing.
- Masking effects are often associated with surfactants, chelating agents, certain proteins, and complex peptide or pharmaceutical formulations. These substances can disrupt the structure of LPS aggregates or shield the lipid A region responsible for activating endotoxin assays.
- When masking occurs, both LAL and rFC assays may fail to detect endotoxin even if contamination is present. As a result, specialized testing strategies—such as hold-time studies, dilution protocols, or orthogonal assays like MAT—are sometimes used to confirm endotoxin status in complex samples [6].
What factors should researchers consider when choosing an LPS detection method?
- Selecting an appropriate LPS detection method depends on several experimental and regulatory factors. Researchers typically evaluate sample composition, required sensitivity, assay specificity, and regulatory expectations.
- For example, samples containing complex matrices, surfactants, or biological materials may require methods that minimize interference or provide broader pyrogen detection. In these cases, MAT may be considered alongside endotoxin-specific assays. For routine endotoxin quantification in relatively simple samples, LAL or rFC assays are commonly used due to their high sensitivity and established validation frameworks.
- Additional considerations include sustainability of assay reagents, compatibility with the sample matrix, regulatory acceptance, and the potential for endotoxin masking. Careful evaluation of these factors helps ensure reliable detection results and improves overall quality control in laboratory workflows [1][3][5].
References
-
Marius M, Vacher F, Bonnevay T, et al. Comparison of Limulus Amebocyte Lysate and Recombinant Factor C Assays for Endotoxin Detection in Four Human Vaccines with Complex Matrices. PDA Journal of Pharmaceutical Science and Technology. 2020;74(4):394–407.
https://pubmed.ncbi.nlm.nih.gov/32179709/ -
Thorne PS, Perry SS, Saito R, et al. Evaluation of the Limulus amebocyte lysate and recombinant factor C assays for assessment of airborne endotoxin. Applied and Environmental Microbiology. 2010;76(15):4988–4995.
https://pubmed.ncbi.nlm.nih.gov/20525858/ -
Iwanaga S. Biochemical principle of Limulus test for detecting bacterial endotoxins. Proceedings of the Japan Academy Series B. 2007;83(4):110–119.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3756735/ -
Solati S, et al. The monocyte activation test detects potentiated cytokine release resulting from the synergistic effect of endotoxin and non-endotoxin pyrogens. Innate Immunity. 2022;28(3–4):130–137.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9136467/ -
Andrade SS, et al. Comparative evaluation of the human whole blood and human peripheral blood monocyte tests for pyrogens. International Journal of Pharmaceutics. 2003;265(1–2):115–124.
https://pubmed.ncbi.nlm.nih.gov/14522124/ -
Kang DH, et al. A Study on the Application of Recombinant Factor C (rFC) Assay Using Biopharmaceuticals. Microorganisms. 2024;12(3):516.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10972375/ -
Abate W, et al. Evaluation of recombinant factor C assay for the detection of divergent lipopolysaccharide structural species and comparison with Limulus amebocyte lysate-based assays and a human monocyte activity assay. Journal of Medical Microbiology. 2017;66(7):888–897.
https://pubmed.ncbi.nlm.nih.gov/28693666/ -
Reich J, Lang P, Grallert H, et al. Masking of endotoxin in surfactant samples: Effects on Limulus-based detection systems. Biologicals. 2016;44(5):417–422.
https://pubmed.ncbi.nlm.nih.gov/27464990/ -
Gorman A, et al. Investigating the effects of chelating agents, surfactants and magnesium cations on the size of LPS aggregates in formulations causing low endotoxin recovery in Limulus amebocyte lysate assays. European Journal of Pharmaceutics and Biopharmaceutics. 2025.
https://pubmed.ncbi.nlm.nih.gov/40925493/ -
Schwarz H, Gornicec J, Neuper T, et al. Biological Activity of Masked Endotoxin. Scientific Reports. 2017;7:44750.
https://pubmed.ncbi.nlm.nih.gov/28317862/
