Endotoxin-Free Peptide Prep Water Quality Standards
Author: Dr. Numan S. Date: February 12, 2026

Learn how water quality impacts peptide preparation and why proper purification standards help reduce endotoxin risk and improve research reliability.
Why Water Quality Matters in Peptide Preparation
In peptide preparation, water is the solvent, diluent, and rinse fluid. If its ionic load or organic carbon drifts, solubility, chromatography baselines, and reaction side-pathways can drift with it [1].
Water is also used “on demand,” so quality has to be engineered into the system, not inferred after the fact. Pharmaceutical guidance highlights controlling quality continuously across generation, storage, and distribution [2].
Types of Laboratory Water Used in Peptide Preparation
Distilled or deionized water can be “chemically improved” yet still fail biological needs. Endotoxin is heat-stable, and water can be re-contaminated by system surfaces and containers; distilled water therefore does not guarantee endotoxin-free water [3].
Ultrapure water standards specify tighter chemical targets. One reagent-water specification defines Type I water at ≤0.0555 µS/cm conductivity (≥18 MΩ·cm resistivity) and ≤50 µg/L TOC (as carbon. An international analytical-lab standard similarly defines Grade 1 water for stringent work and notes it is typically produced by further treatment plus fine filtration. These specifications are laboratory water standards used to define ultrapure water in day-to-day research workflows. [1]
Key Water Quality Parameters to Monitor
For peptide solutions, a practical minimum set is conductivity/resistivity, TOC, and particulate control (final filtration). These are central to laboratory water standards and provide rapid signals that water quality has changed before experiments do. Water quality testing is strongest when these signals are trended, not just spot-checked. [1]
Endotoxin and microbes are separate from ions. A study of commercial waters found some waters inhibited chromogenic Limulus amebocyte lysate measurements due to trace metals, so water quality testing must consider assay interference, not only purity labels [4].
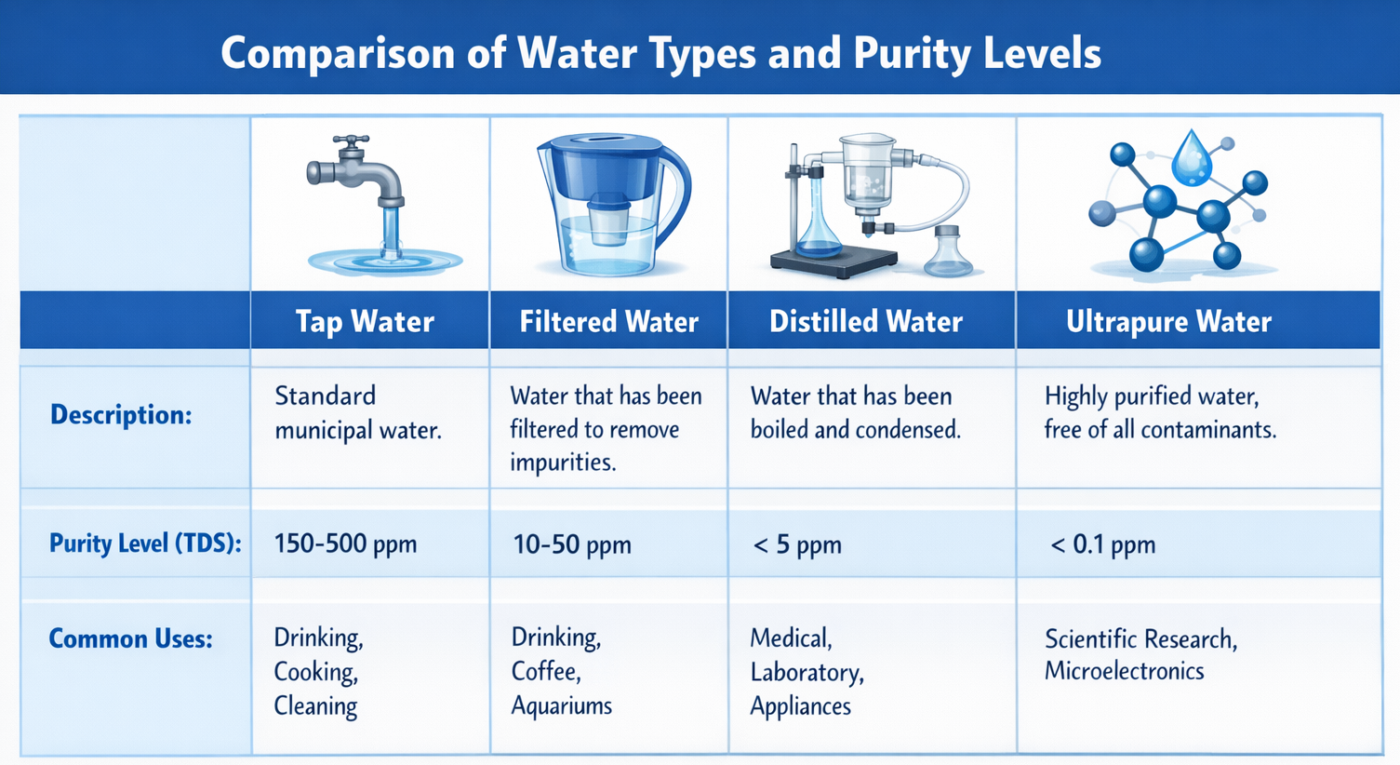
Figure 1: Comparison of Water Types and Purity Levels infographic
Metal-catalyzed oxidation is a concrete mechanism: experiments show methionine-containing peptides undergo metal-driven oxidation through radical chemistry in Fenton-type systems. Keeping metal ions low is therefore a stability control, not just an analytical preference [5].
Endotoxin contamination can also create false biological “hits.” A screen of common research reagents reported lipopolysaccharide in multiple commercial preparations, including synthetic peptides, and showed that LPS can drive cellular activation that looks like true activity [6].
Storage and Handling of Laboratory Water
High-purity water does not stay “high-purity” automatically. An analytical-lab standard warns that conductivity targets apply to freshly prepared water and can shift during storage due to atmospheric carbon dioxide and container leaching. Short hold times are therefore part of good peptide preparation. [7]
At the system level, guidance emphasizes routine monitoring and controls to minimize microbial growth and endotoxin formation in storage and distribution loops. At the bench, that translates to clean dedicated bottles, short hold times, and no “top-offs.” [2]
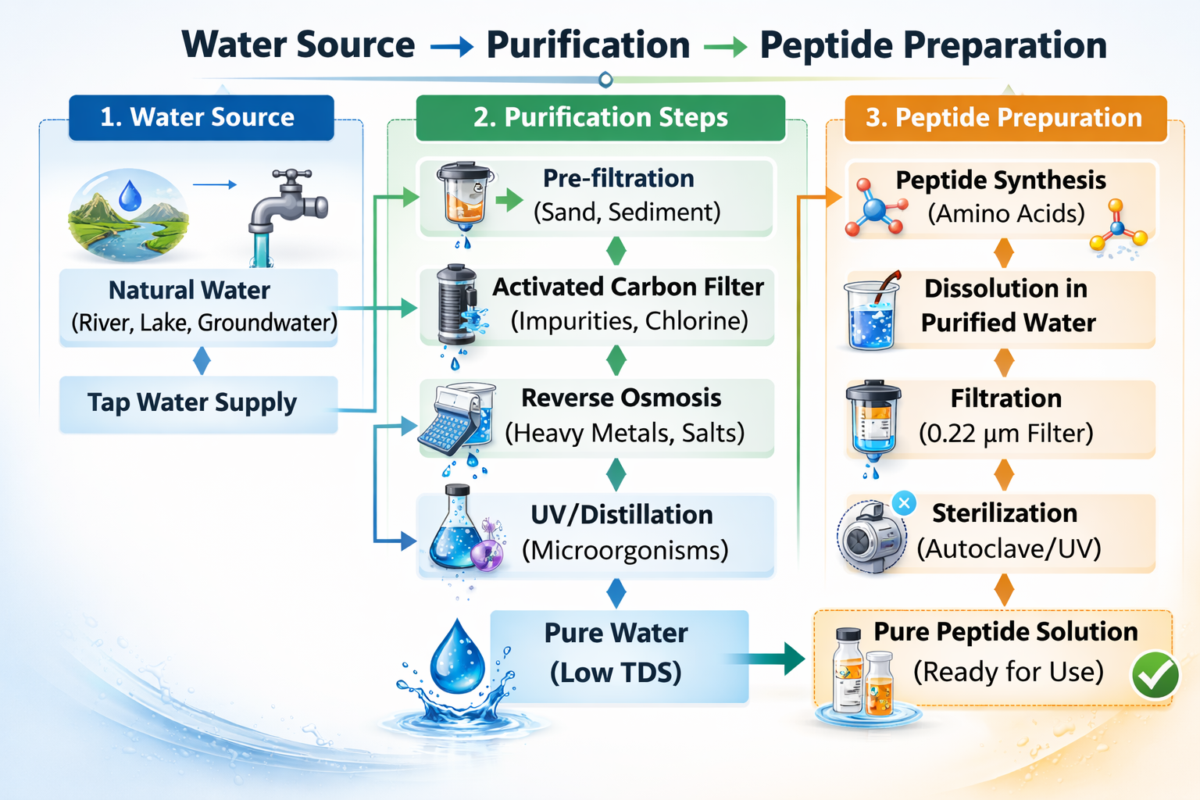
Figure 2: Water purification for peptide preparation.
Best Practices for Endotoxin-Conscious Peptide Preparation
Reconstitution should match downstream biology. For cell-based work, choose endotoxin-free water that is specified and verified for low endotoxin (ie, below a defined limit or assay detection threshold), and treat consumables as potential endotoxin sources unless qualified [8].
Document what matters. Log the water grade, source/dispense point, and time-to-use for each peptide preparation, plus outlet conductivity/resistivity (and TOC when available), so failures can be traced to controllable inputs and reconciled with water quality testing records [1].
Common Misconceptions About “Clean” Water
Sterile is not the same as depyrogenated. A kinetic analysis summarizes that a 3‑log endotoxin reduction by dry heat typically requires roughly 250°C for 30 minutes, illustrating why destroying endotoxin is harder than killing bacteria [9].
Likewise, “distilled” is not synonymous with endotoxin-free water, and negative results are not definitive if the method is inhibited by matrix effects or assay water itself. If results are inconsistent, treat endotoxin contamination as a hypothesis you can test [4].
High-Quality Water Is Foundational to Reliable Peptide Preparation
High-quality water underpins reliable peptide preparation: apply laboratory water standards for chemical control, add verified endotoxin control when biology demands it, and manage storage/handling as part of the purification system [10].
Frequently asked questions (FAQs) about Endotoxin-Free Peptide Prep Water Quality
What type of water should be used for peptide preparation?
- For most research-grade peptide preparation, Type I ultrapure water is recommended. Type I water typically has a resistivity of 18.2 MΩ·cm at 25°C, total organic carbon (TOC) levels below 5–10 ppb, and extremely low microbial and endotoxin content.
- In highly sensitive applications—such as cell culture studies, injectable formulation development, or endotoxin-sensitive bioassays—Water for Injection (WFI) or validated endotoxin-controlled ultrapure water systems may be required. The choice depends on the downstream application, regulatory context, and acceptable endotoxin limits.
- Distilled or deionized (DI) water may be acceptable for basic chemical handling, but they are generally insufficient for endotoxin-sensitive peptide research unless further purified and validated.
How does water quality affect endotoxin levels?
- Endotoxins (lipopolysaccharides, LPS) originate from Gram-negative bacteria and can persist even after bacteria are no longer viable. Poor-quality water systems can harbor biofilms within storage tanks, tubing, or filters, leading to continuous endotoxin shedding into the water supply.
- If such water is used during peptide reconstitution, dilution, or purification, endotoxin contamination can be introduced despite otherwise sterile technique. Importantly, endotoxins are heat-stable and not removed by simple sterilization alone.
- Therefore, water purity directly influences endotoxin load, especially in workflows involving lyophilized peptides, injectable research preparations, or in vitro immune assays.
Is distilled water sufficient for sensitive peptide work?
- In most cases, no. While distilled water removes many ionic contaminants and some microorganisms through boiling and condensation, it does not guarantee low endotoxin levels. Endotoxins can survive distillation under certain conditions and may be reintroduced during storage.
- Additionally, distilled water systems are often not continuously monitored for TOC, resistivity, or endotoxin burden. For sensitive peptide applications—such as receptor binding studies, cytokine assays, or animal research—distilled water alone is typically inadequate unless further processed through validated ultrapure systems.
What are the standards for ultrapure laboratory water?
Type I ultrapure laboratory water typically meets the following benchmarks:
- Resistivity: 18.2 MΩ·cm at 25°C
- Total Organic Carbon (TOC): <5–10 ppb
- Bacterial count: <1 CFU/mL
- Endotoxin level: Often <0.03–0.25 EU/mL (depending on system and validation protocol)
High-quality systems use multiple purification stages, including reverse osmosis, deionization, activated carbon filtration, UV oxidation, ultrafiltration, and sub-micron membrane filtration.
For peptide preparation used in pharmaceutical development contexts, compliance with USP, EP, or equivalent pharmacopeial standards may be required.
How can poor water quality impact research results?
Poor water quality can affect peptide research in several ways:
- Inflammatory artifacts: Endotoxin contamination can activate immune cells, skewing cytokine data or falsely elevating inflammatory markers.
- Protein/peptide instability: Dissolved organic compounds or metal ions may promote oxidation, aggregation, or degradation.
- Reproducibility issues: Variable microbial or endotoxin burden can introduce batch-to-batch inconsistency.
- False assay signals: Endotoxin contamination can interfere with bioassays, receptor studies, and in vivo models.
Even trace contamination can significantly alter outcomes in sensitive systems, particularly when studying immunomodulatory peptides or conducting cell-based experiments.
References
- ASTM International. Standard Specification for Reagent Water (D1193-24). ASTM International; 2024.
- International Organization for Standardization. Water for Analytical Laboratory Use—Specification and Test Methods (ISO 3696:1987). ISO; 1987.
- World Health Organization. Good Manufacturing Practices: Water for Pharmaceutical Use. WHO Technical Report Series No. 1033, Annex 3. World Health Organization; 2021.
- Li L, Menon A, Tanaka T, et al. Kinetics of hydrothermal inactivation of endotoxins. Appl Environ Microbiol. 2011;77(11):3738-3745.
- Weinstein JR, Quan Y, Hanson JF, et al. Lipopolysaccharide is a frequent and significant contaminant in microglia-activating factors. Glia. 2008;56(1):16-26.
- Dunér KI, Ragnarsson U. The importance of the quality of water in Limulus amebocyte lysate tests. Pharm Acta Helv. 1995;70(5):321-324.
- Hong J, Schöneich C. The metal-catalyzed oxidation of methionine in peptides by Fenton systems involves two consecutive one-electron oxidation processes. Free Radic Biol Med. 2001;31(11):1432-1441.
