Endotoxin Load Monitoring Throughout Peptide Workflows
Author: Dr. Numan S. Date: April 16, 2026

Why Endotoxin Load Matters in Peptide Workflows
Endotoxin load refers to the total amount of bacterial endotoxin present within a material, process stream, or finished sample. In peptide workflows, endotoxins are commonly associated with lipopolysaccharides from Gram-negative bacteria that can persist on surfaces, in water systems, and within raw materials.[1] Because endotoxins are heat-stable and difficult to remove completely, controlling endotoxin load is an important part of maintaining reliable laboratory conditions.
Monitoring endotoxin load matters because contamination can affect analytical consistency, purification performance, and downstream handling processes.[2] Laboratories that implement structured endotoxin monitoring programs are often better positioned to identify contamination sources early and reduce unnecessary process variation during peptide manufacturing and handling.
How Endotoxin Load Can Change Across a Peptide Workflow
Endotoxin load does not remain constant throughout peptide workflows. Levels may increase during synthesis, cleavage, purification, filtration, lyophilization, or sample transfer if environmental controls are insufficient.[3] Even routine handling steps can introduce endotoxins through contact with contaminated tools, containers, or water systems.
Changes in endotoxin load are often influenced by process complexity and operator interaction. Multiple transfers between vessels or poorly controlled storage conditions can increase contamination risk over time.[4] Monitoring throughout each workflow stage helps identify where endotoxin accumulation may occur and where corrective actions are most effective.
How Purification and Handling Steps Affect Endotoxin Load
Purification methods can significantly influence endotoxin load during peptide production. Techniques such as reversed-phase chromatography, ultrafiltration, and affinity purification may reduce endotoxin content when properly optimized.[9] However, purification equipment itself can become a contamination source if cleaning validation is inadequate.
Handling practices are equally important because repeated exposure to open environments increases contamination opportunities. Improper storage containers, excessive sample transfers, and inconsistent aseptic procedures may elevate endotoxin load even after successful purification.[6] Standardized handling protocols are therefore important for maintaining cleaner peptide processing conditions.
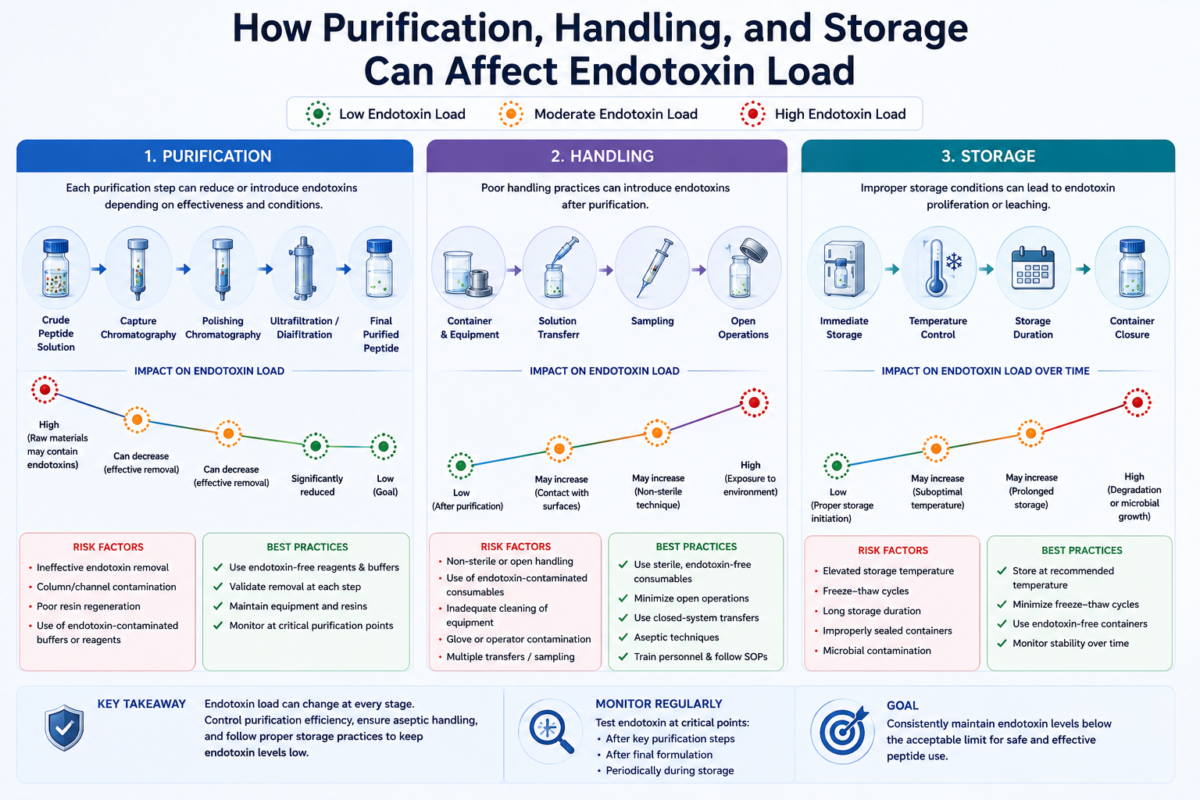
Figure 1: Endotoxin load control in peptide production.
Several analytical methods are used to measure endotoxin load in laboratory settings. Limulus Amebocyte Lysate assays remain among the most widely used techniques because of their sensitivity and regulatory acceptance.[10] Gel clot, chromogenic, and turbidimetric formats are commonly applied depending on workflow requirements and detection limits.
Recombinant Factor C assays are also increasingly used for endotoxin monitoring because they target endotoxin-specific activation pathways while avoiding certain biological variability concerns associated with traditional lysate-based assays.[11] Regardless of the method selected, assay validation is necessary to confirm reliable performance within specific peptide matrices.
How Endotoxin Load Is Monitored During Peptide Production
Endotoxin monitoring typically involves scheduled testing at predefined process stages. Samples may be collected from purified water systems, synthesis intermediates, equipment rinse solutions, or final peptide preparations to evaluate endotoxin load over time.[7] Trend analysis can help laboratories identify recurring contamination patterns and improve process consistency.
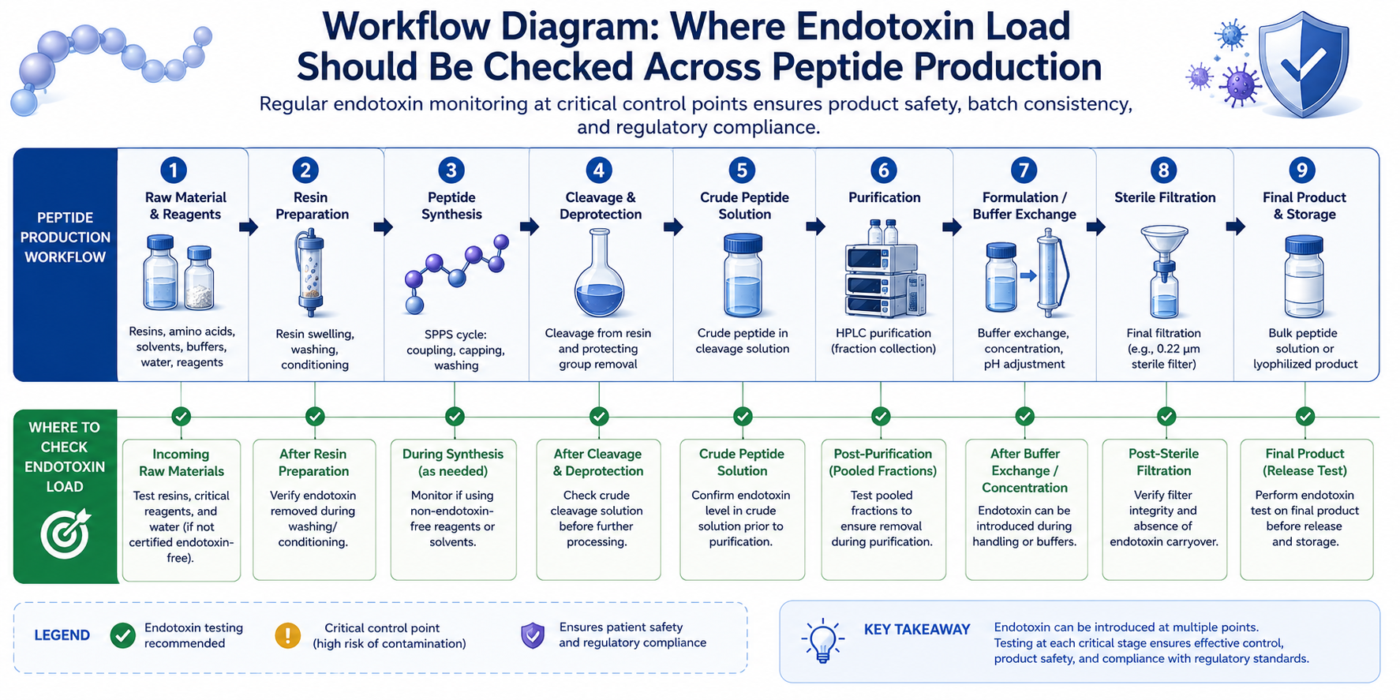
Figure 2: Peptide production endotoxin testing workflow
Routine endotoxin monitoring often relies on validated assay methods capable of detecting low endotoxin concentrations. Testing frequency varies depending on workflow complexity, production scale, and quality objectives.[8] Facilities that maintain consistent monitoring schedules are generally more effective at detecting process deviations before they affect final product handling.
Why Sampling Points Matter for Endotoxin Load Tracking
Sampling locations strongly influence the accuracy of endotoxin load assessments. Testing only final materials may overlook contamination introduced earlier in the process. For this reason, many laboratories establish multiple sampling points throughout peptide workflows to improve traceability and process understanding.[2]
Strategic sampling before and after purification, filtration, or storage steps allows researchers to evaluate whether endotoxin load is increasing or decreasing at critical stages.[5] Effective sampling plans also help determine whether contamination originates from equipment, water systems, handling procedures, or environmental exposure.
What Sources Contribute to Endotoxin Load?
Several factors contribute to endotoxin load during peptide production. Water systems are among the most common sources because bacterial growth within improperly maintained purification systems can release endotoxins into process water.[5] Raw materials, filtration membranes, glassware, and reusable equipment may also contribute contamination if cleaning procedures are inconsistent.
Environmental exposure is another important factor. Airborne particles, laboratory surfaces, and operator contact can all influence endotoxin load within open processing environments.[6] Cleanroom practices, gowning procedures, and controlled airflow systems are commonly used to minimize microbial and endotoxin exposure during sensitive production activities.
Common Challenges in Endotoxin Load Monitoring
One major challenge in endotoxin load monitoring is assay interference caused by solvents, buffers, detergents, or peptide formulations.[8] Certain sample components may inhibit or enhance assay responses, making accurate interpretation more difficult without appropriate controls and validation studies.
Another challenge involves maintaining consistent monitoring practices across long production cycles. Variability in sampling technique, environmental conditions, or equipment sanitation can affect peptide quality and reduce data reliability.[4] Strong documentation systems, operator training, and preventive maintenance programs are important for supporting a more reliable endotoxin monitoring process.
Frequently asked questions (FAQs) about Endotoxin Load Monitoring Throughout Peptide Workflows
What is endotoxin load?
- Endotoxin load refers to the total amount of bacterial endotoxins, primarily lipopolysaccharides (LPS) from Gram-negative bacteria, present within a peptide sample, solution, surface, or production environment. Endotoxin load may accumulate throughout peptide workflows due to contaminated raw materials, water systems, equipment contact surfaces, or handling practices. Because endotoxins can persist even after bacteria are removed or destroyed, monitoring total endotoxin burden remains an important part of peptide quality assessment.
Why does endotoxin load matter throughout peptide workflows?
- Endotoxin load matters because contamination can affect analytical consistency, workflow reliability, and overall peptide quality. Elevated endotoxin levels may interfere with sensitive laboratory assays, compromise sterility-focused workflows, and increase variability during downstream processing. Since endotoxin contamination can occur at multiple stages of synthesis, purification, filtration, storage, and reconstitution, maintaining low endotoxin load throughout the entire workflow helps support cleaner and more reproducible peptide preparation processes.
How is endotoxin load monitored during peptide production?
- Endotoxin load is commonly monitored using endotoxin detection assays such as Limulus Amebocyte Lysate (LAL)-based methods or recombinant Factor C (rFC) testing. Sampling may occur at multiple workflow stages, including after synthesis, purification, filtration, lyophilization, and final packaging. Laboratories may also monitor endotoxin load indirectly through environmental monitoring programs, water quality testing, equipment validation, and contamination trend analysis. Routine testing helps identify where endotoxin introduction may be occurring within the production process.
What factors can increase endotoxin load across a workflow?
- Several factors can contribute to increased endotoxin load during peptide workflows. Common sources include contaminated water systems, poorly cleaned equipment, inadequate environmental controls, repeated material transfers, improper storage conditions, and excessive operator handling. Endotoxins may also accumulate when biofilms develop in fluid pathways or when filtration systems are not properly maintained. In some workflows, insufficient depyrogenation procedures or cross-contamination between batches can further elevate endotoxin burden.
What best practices help reduce endotoxin load and support peptide quality?
- Reducing endotoxin load typically requires a combination of contamination control strategies across the full peptide workflow. Common best practices include using endotoxin-controlled water, implementing validated cleaning and depyrogenation procedures, minimizing unnecessary handling steps, maintaining cleanroom environmental controls, and regularly testing high-risk workflow stages. Proper storage conditions, sterile consumables, and routine equipment maintenance may also help reduce contamination risk. Consistent endotoxin monitoring throughout production supports better process control and helps maintain peptide quality standards.
References
- Petsch D, Anspach FB. Endotoxin removal from protein solutions. J Biotechnol. 2000;76(2-3):97-119.
https://pubmed.ncbi.nlm.nih.gov/10656326/ - Magalhães PO, Lopes AM, Mazzola PG, et al. Methods of endotoxin removal from biological preparations: a review. J Pharm Pharm Sci. 2007;10(3):388-404.
https://pubmed.ncbi.nlm.nih.gov/18078000/ - Gorbet MB, Sefton MV. Endotoxin: the uninvited guest. Biomaterials. 2005;26(34):6811-6817.
https://pubmed.ncbi.nlm.nih.gov/15939467/ - Dubczak J, Korycka-Machała M, Rumijowska-Galewicz A, et al. Strategies for reducing endotoxin contamination in pharmaceutical production. Molecules. 2021;26(19):5967.
https://www.mdpi.com/1420-3049/26/19/5967 - International Organization for Standardization. ISO 3696: Water for Analytical Laboratory Use—Specification and Test Methods. ISO; 1987.
https://www.iso.org/standard/9169.html - Whyte W. Cleanroom Technology: Fundamentals of Design, Testing and Operation. 3rd ed. Wiley-Blackwell; 2023.
https://www.wiley.com/en-us/Cleanroom+Technology%3A+Fundamentals+of+Design%2C+Testing+and+Operation%2C+3rd+Edition-p-9781119776805 - United States Pharmacopeia. USP <85> Bacterial Endotoxins Test. United States Pharmacopeial Convention; 2024.
https://doi.org/10.31003/USPNF_M98830_02_01 - Bolden J, Smith K. Assay interference and inhibition in bacterial endotoxin testing. PDA J Pharm Sci Technol. 2017;71(5):396-405.
https://pubmed.ncbi.nlm.nih.gov/28814468/ - Ongkudon CM, Chew JH, Liu B, Danquah MK. Chromatographic removal of endotoxins: a bioprocess engineer’s perspective. ISRN Chromatogr. 2012;2012:649746.
https://pmc.ncbi.nlm.nih.gov/articles/PMC3356978/ - Levin J, Bang FB. The role of endotoxin in the extracellular coagulation of Limulus blood. Bull Johns Hopkins Hosp. 1964;115:265-274.
https://pubmed.ncbi.nlm.nih.gov/14204998/ - Maloney T, Phelan R, Simmons N. Saving the horseshoe crab: a synthetic alternative to horseshoe crab blood for endotoxin detection. PLoS Biol. 2018;16(10):e2006607.
https://journals.plos.org/plosbiology/article?id=10.1371/journal.pbio.2006607 - United States Pharmacopeia. USP <86> Bacterial Endotoxins Test Using Recombinant Reagents. United States Pharmacopeial Convention; 2024.
https://doi.org/10.31003/USPNF_M16015_02_01
