Endotoxin Masking and Interference in Peptide Samples
Author: Dr. Numan S. Date: February 5, 2026

Learn how endotoxin masking can interfere with detection in samples, why it happens, and best practices used to reduce interference risks in lab testing. In practice, endotoxin masking can make a peptide-containing matrix appear “clean” while biologically active endotoxin is still present—an outcome that matters for both product safety and experimental interpretation [1], [2], [6].
What Is Endotoxin Masking?
Masking describes a loss of assayable endotoxin signal (often time dependent) even when endotoxin molecules have not been physically removed from the sample. This is widely discussed under the “low endotoxin recovery” (LER) umbrella because spiked endotoxin standards may become difficult to detect under certain matrix conditions. [2]
A key implication is that endotoxin assays, including the most common compendial formats, are measuring a functional response (bioactivity in the assay system), not simply the mass of lipopolysaccharide present. When the supramolecular state of endotoxin changes, the measured signal can drop even though endotoxin remains in solution.[3]
Endotoxins are lipopolysaccharides from Gram-negative bacteria; LPS endotoxin is among the most potent inflammatory triggers encountered in biomedical contexts. In controlled settings, very low intravenous exposures can provoke measurable inflammatory responses in humans, underscoring why tight control is required for parenteral products and why background contamination can derail mechanistic biology. [4]
From a laboratory standpoint, endotoxin can enter workflows through raw materials, water, consumables, and research-grade biologics/peptides that are not screened to pharmaceutical standards. A systematic screen of common research reagents found that some commercial protein and synthetic peptide preparations contained detectable LPS, reinforcing that “unintended endotoxin” is not a rare edge case. [5]
What Are Endotoxins and Why Detection Is Important
Endotoxins are lipopolysaccharides from Gram-negative bacteria; LPS endotoxin is among the most potent inflammatory triggers encountered in biomedical contexts. In controlled settings, very low intravenous exposures can provoke measurable inflammatory responses in humans, underscoring why tight control is required for parenteral products and why background contamination can derail mechanistic biology [4].
From a laboratory standpoint, endotoxin can enter workflows through raw materials, water, consumables, and “research-grade” biologics/peptides that are not screened to pharmaceutical standards.A systematic screen of common research reagents found that some commercial protein and synthetic peptide preparations contained detectable LPS, reinforcing that “unintended endotoxin” is not a rare edge case [5].
Endotoxin Masking vs Endotoxin Interference
In peptide-rich matrices, one well-supported mechanism is electrostatic and amphiphilic binding: cationic peptides and proteins can bind the anionic lipid A region of LPS and reduce its accessibility to Limulus-based reagents, lowering measured recovery. This binding-driven loss of signal is functionally consistent with false-negative readouts—especially when the peptide is present at concentrations that favor complex formation [6].

Figure 1: Endotoxin interference in Peptide Analysis
This is not only a conceptual concern; experimental work in blood/serum systems explicitly notes that LAL results can shift toward false negatives when LPS-binding polypeptides remain present as inhibitors that are not eliminated by routine heat/dilution pretreatments. Although peptide products differ from plasma, the core biophysical point carries over: peptide–LPS association can reduce detectability if the assay depends on LPS being presented in an activation-competent form [7].

Interference is a broad analytical category: endotoxin interference can occur when sample components inhibit or enhance the enzymatic cascade, shift pH/ionic conditions, drive adsorption, or distort optical readouts in photometric formats. The regulatory and compendial ecosystem explicitly treats interfering factors as a core validation concern (eg, confirming labeled lysate sensitivity and checking for inhibiting/enhancing factors in the sample matrix) [8].
By contrast, masking (as studied in LER contexts) is often framed as a change in endotoxin’s assayable state over time (eg, under chelator/surfactant exposure) that can persist even when the sample “passes” immediate inhibition/enhancement checks. In other words, endotoxin interference can be an assay chemistry problem, while masking can be an endotoxin–matrix state problem that manifests as delayed signal loss [9].
How Masking Affects Endotoxin Testing Methods (High-Level)
Most routine workflows rely on common BET formats—gel-clot and photometric (turbidimetric/chromogenic) techniques—built around Limulus lysate activation in the presence of endotoxin. Because these assays are biochemical cascades and/or optical readouts, they are vulnerable to assay interference from pH shifts, chelators, viscous materials, adsorption, and product color/turbidity that can suppress or distort the measurable endpoint [8].
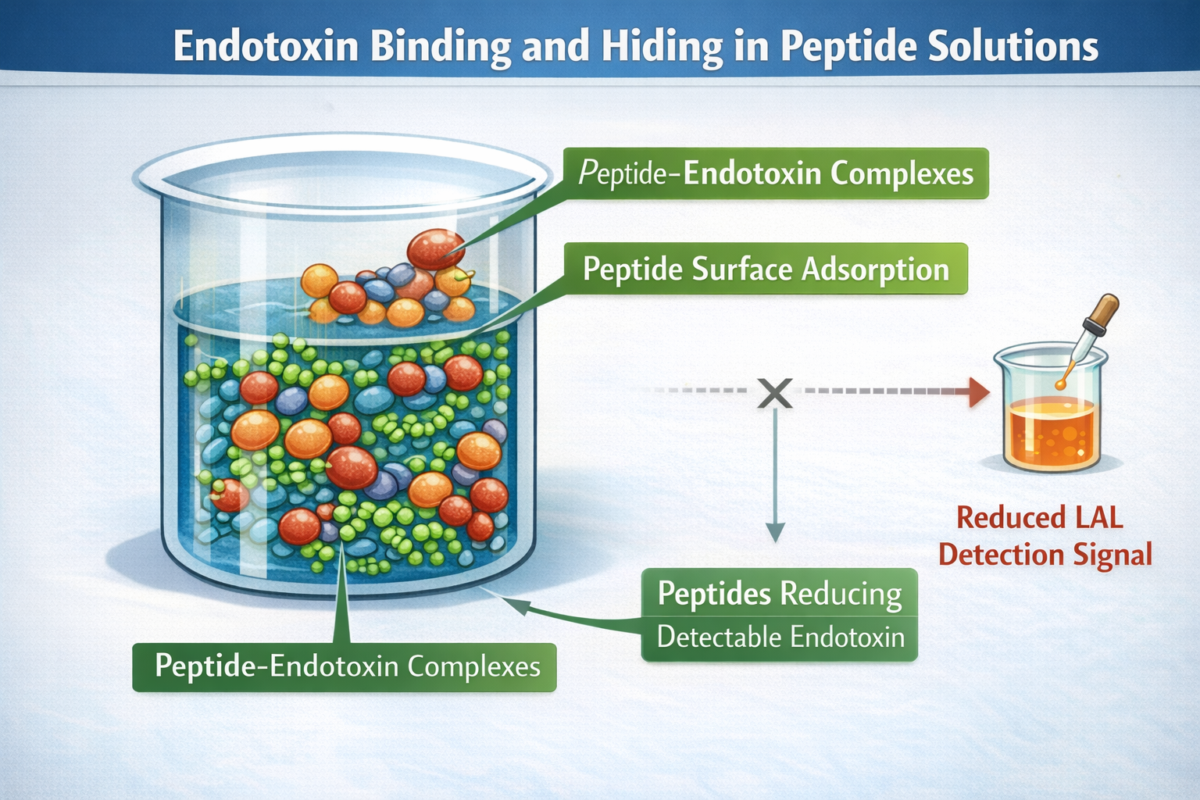
Figure 3:Endotoxin binding in peptide solutions
Mechanistically, one proposed reason masking can reduce signal is that activation requires endotoxin to be presented in a surface/aggregate context that supports Factor C activation; when matrix conditions decrease the effective activating surface, signal falls. This is consistent with reports emphasizing that LER reflects changes in endotoxin activity state rather than merely “blocking the reaction,” highlighting why the same endotoxin amount can yield different assay outputs depending on how it is organized in solution [12].
Compendial practice places strong emphasis on suitability/validity checks (including interfering factors testing), yet regulators also caution that sample storage and handling can affect detectability and that stability of assayable endotoxin should be supported by data.^6 Put bluntly, a sample can test low at one timepoint and still contain endotoxin later measured as “missing” because the assay response has diminished during the hold period—an archetypal false-negative path in endotoxin testing [13].
Risk Factors That Increase the Likelihood of Masking
Several matrix features recur across published discussions: chelators can remove divalent cations that stabilize LPS aggregate structure, while surfactants can reorganize endotoxin into forms less accessible to Limulus-based detection. In plasma studies, citrate anticoagulation markedly reduced recovery compared with aqueous controls, supporting the general principle that divalent-cation chemistry can shift assayable activity [14].
A second risk cluster is molecular binding and adsorption. Cationic peptides/proteins can bind LPS and hinder LAL activation, and physical effects such as endotoxin adsorption to surfaces or high viscosity can also suppress readout. For peptide-rich solutions, this combination means that both peptide–LPS complexation and container interactions are credible contributors to under-recovery when conditions favor binding or surface loss [15].
One strategy is explicitly time-aware: conduct stability/hold-time evaluations in the actual matrix, because regulators note that detectability can shift with storage/handling and that protocols should consider endotoxin source and native-versus-purified behavior. Reich and colleagues further emphasize that susceptibility can depend on the endotoxin source and cannot be reliably predicted a priori, which is why selecting an appropriate spike and study design matters [13].
A second strategy is orthogonality. Recombinant Factor C–based methods remove some lysate-derived pathways/impurities and can provide comparable performance to LAL in multi-year proficiency testing, with reports of equal or improved recovery in certain settings. This does not “solve” every matrix problem, but it can reduce confounding pathways and offer a complementary lens when LAL-based results are questionable [16].
Best Practices in Handling Peptide Samples
At the workflow level, peptide sample handling should treat endotoxin control as both a contamination-prevention problem and a detectability problem. Harmonized pharmacopeial text stresses depyrogenation of heat-stable materials and emphasizes performing preparatory checks such as labeled lysate sensitivity confirmation and tests for interfering factors, reflecting how foundational handling and validation steps are to reliable outcomes [17].
Equally important, peptide sample handling includes minimizing conditions known to shift assayable endotoxin: avoid unnecessary hold times at problematic temperatures, ensure adequate mixing (to avoid nonrepresentative aliquots), and document stability of assayable endotoxin under the same storage/handling conditions used operationally. These practices directly target the question, “Can a sample test low for endotoxin but still contain it?”—yes, if detectability changes after sampling [18].
A frequent misconception is equating “passing” a single run’s controls with “no endotoxin.” Regulatory technical guidance notes that excessive dilution can make a negative result meaningless because endotoxin may simply be diluted below the detection limit; thus, a seemingly clean result can still coexist with clinically relevant contamination if testing conditions push beyond valid interpretive bounds [19].
Another misconception is that masking is only relevant to large biologics. The literature on LER emphasizes that matrix features (chelators, surfactants, and other excipients) and LPS supramolecular behavior are central drivers of under-detection—features that can also appear in peptide formulations and peptide-processing buffers [20].
Why Understanding Masking Improves Research Reliability
In research biology, endotoxin contamination is a well-documented confounder: peptide and protein reagents can carry LPS that triggers innate immune pathways, creating false mechanistic conclusions if not controlled. Recognizing masking/interference risks strengthens experimental design by motivating routine endotoxin measurement, appropriate controls (including polymyxin-sensitive checks when relevant), and skepticism toward unexpected inflammatory phenotypes driven by reagent impurities [5].
Because both matrix-driven state changes (LER-style) and direct inhibition/enhancement mechanisms can create false negatives, the safest operational stance is to treat endotoxin masking as a plausible hidden variable whenever peptide matrices, surfactants/chelators, or complex biological additives are involved. This framing directly answers “Why can endotoxin tests give false negatives?”—sometimes the assay is not wrong; it is reporting reduced assayable endotoxin under the tested conditions [9].
Frequently asked questions (FAQs) about Endotoxin Masking
What is endotoxin masking and why is it a concern in peptide samples?
- Endotoxin masking refers to a phenomenon in which lipopolysaccharide (LPS) from Gram-negative bacteria becomes partially or fully undetectable in standard endotoxin assays, despite being present in the sample. This typically occurs when LPS interacts with formulation components—such as peptides, surfactants, chelators, or buffers—that alter its aggregation state or reduce its biological activity in the assay system.
- In peptide samples, masking is a concern because many synthetic or recombinant peptides possess amphipathic or cationic properties that can bind to LPS. When LPS is bound or structurally altered, it may not efficiently activate Limulus amebocyte lysate (LAL) or recombinant factor C (rFC) assays. The result is a false-negative or artificially low endotoxin reading. In research and pharmaceutical development settings, this can compromise quality control and risk assessments.
How can peptides interfere with endotoxin detection assays?
Peptides can interfere with endotoxin detection through several mechanisms:
- Direct binding to LPS: Cationic or amphiphilic peptides may bind to the negatively charged lipid A region of endotoxin, altering its accessibility to detection reagents.
- Disruption of LPS aggregates: Endotoxin naturally forms micellar or aggregated structures in aqueous environments. Certain peptides can change these aggregate states, reducing effective activation of assay components.
- Assay inhibition: Some peptides may inhibit enzymatic reactions in LAL-based assays, particularly gel-clot or chromogenic formats, by altering pH, ionic strength, or enzyme kinetics.
- Protein-binding effects: Peptides in complex matrices can sequester endotoxin in a way that limits interaction with factor C, the primary LPS-sensing component in both traditional LAL and recombinant assays.
Because endotoxin assays rely on biological recognition of lipid A, any factor that reduces LPS accessibility or alters its conformation can interfere with detection.
Why might endotoxin levels appear falsely low in lab tests?
Endotoxin levels may appear falsely low for several reasons:
- Masking or sequestration: As described above, LPS may be present but biologically inactive in the assay due to binding or structural modification.
- Inadequate sample preparation: Insufficient dilution, lack of validated hold-time studies, or improper spike recovery testing can lead to incomplete detection.
- Matrix effects: High peptide concentrations, salts, detergents, or chelating agents (e.g., EDTA) can interfere with assay chemistry.
- Time-dependent masking: In some cases, endotoxin becomes progressively masked after storage in certain buffer systems, leading to declining detectable levels over time.
For this reason, regulatory and quality frameworks recommend performing inhibition/enhancement controls and spike recovery studies to confirm assay suitability for each peptide formulation.
What lab practices help reduce endotoxin masking risk?
Several best practices can reduce the risk of endotoxin masking in peptide workflows:
- Perform validated spike recovery testing: Add a known endotoxin standard to the peptide matrix to confirm that the assay can accurately recover it.
- Evaluate dilution strategies: Dilution may reduce matrix interference while maintaining detectable endotoxin levels.
- Control formulation components: Avoid unnecessary surfactants, high chelator concentrations, or extreme pH conditions that promote LPS structural changes.
- Conduct hold-time studies: Assess whether endotoxin detectability changes over storage time in specific peptide solutions.
- Use orthogonal detection methods: When appropriate, compare traditional LAL with recombinant factor C assays to identify assay-specific interference.
- Maintain strict contamination control: Prevent introduction of new endotoxin during handling, which can complicate interpretation of masking versus contamination.
These steps help ensure that measured endotoxin levels reflect true sample content rather than assay artifacts.
How does endotoxin masking impact research reliability?
Endotoxin masking can significantly affect research reliability. If endotoxin contamination is present but undetected, downstream biological experiments may produce misleading results. For example:
- Cell culture systems may exhibit unexpected inflammatory responses.
- Animal studies may show immune activation unrelated to the intended peptide mechanism.
- Reproducibility between laboratories may decline if different assay formats detect endotoxin differently.
In peptide research and early-stage pharmaceutical development, unrecognized endotoxin exposure can confound mechanistic interpretation, skew dose-response findings, and undermine translational validity. Ensuring accurate endotoxin detection—free from masking or interference—is therefore essential for maintaining scientific rigor, data integrity, and overall quality assurance.
References
- Reich J, Weyer FA, Tamura H, Nagaoka I, Motschmann H. Low Endotoxin Recovery—Masking of Naturally Occurring Endotoxin. Int J Mol Sci. 2019;20(4):838. doi:10.3390/ijms20040838.
- Gorman A, Golovanov AP. Lipopolysaccharide Structure and the Phenomenon of Low Endotoxin Recovery. Eur J Pharm Biopharm. 2022;180:289-307. doi:10.1016/j.ejpb.2022.10.006.
- Tsuchiya M. Mechanism of Low Endotoxin Recovery Caused by a Solution Containing a Chelating Agent and a Detergent. Immunome Res. 2019;15:166. doi:10.4172/1745-7580.1000166.
- Harm S, Schildböck C, Cont D, et al. Heparin enables the reliable detection of endotoxin in human serum samples using the Limulus amebocyte lysate assay. Sci Rep. 2024;14:2410. doi:10.1038/s41598-024-52735-8.
- Piehler M, Röder R, Blessing S, Reich J. Comparison of LAL and rFC Assays—Participation in a Proficiency Test Program between 2014 and 2019. Microorganisms. 2020;8(3):418. doi:10.3390/microorganisms8030418.
- U.S. Food and Drug Administration. Guidance for Industry: Pyrogen and Endotoxins Testing—Questions and Answers. June 2012.
- European Medicines Agency; International Council for Harmonisation. ICH Q4B Annex 14: Bacterial Endotoxins Tests—General Chapter (Step 3). 2010.
- Weinstein JR, Swarts S, Bishop C, et al. Lipopolysaccharide is a Frequent and Significant Contaminant in Microglia-Activating Factors. J Neuroinflammation. 2008;5:4.
