Future Trends: AI and Automation in Peptide Quality Control
Author: Dr. Numan S. Date: April 23, 2026

What Is Peptide Quality Control?
Peptide quality control refers to the analytical and procedural systems used to verify peptide identity, purity, composition, and consistency before research applications. Modern peptide quality control commonly includes chromatographic testing, mass spectrometry, sterility assessment, and endotoxin screening.[1] These procedures help laboratories confirm that synthesized peptides meet predefined analytical specifications.
Reliable peptide quality control is important because peptide synthesis can introduce impurities such as truncated amino acid sequences, oxidation products, and residual reagents.[2] Analytical verification reduces variability between batches and supports reproducible laboratory outcomes. High-performance liquid chromatography (HPLC) and mass spectrometry remain central tools in peptide analysis because they provide molecular-level characterization of peptide samples.[3].
Why Peptide Quality Control Is Evolving
The increasing complexity of peptide manufacturing has driven significant changes in peptide quality control systems. Research laboratories now process larger datasets generated by advanced analytical instruments, including ultra-high-performance liquid chromatography and high-resolution mass spectrometry.[4] Manual review of these datasets can slow laboratory operations and increase the risk of interpretation inconsistencies.
At the same time, peptide manufacturing workflows require greater precision and documentation. Regulatory expectations for analytical traceability and data integrity have encouraged laboratories to adopt more digitalized systems for quality management.[5] These demands have accelerated interest in AI and automation technologies capable of improving consistency and reducing repetitive manual tasks.
Modern laboratories also require faster turnaround times for analytical verification. Automated systems can streamline sample preparation, data acquisition, and reporting steps that previously required extensive technician involvement.[6] As peptide production scales increase, more facilities are exploring AI and automation to improve operational efficiency without compromising analytical standards.
How AI Could Support Peptide Quality Control
Artificial intelligence has become increasingly relevant in peptide quality control because machine learning systems can analyze large analytical datasets rapidly. AI-based software may assist with chromatographic peak identification, impurity classification, and spectral interpretation during peptide analysis.[7] These systems can help reduce review times while improving consistency across analytical batches.
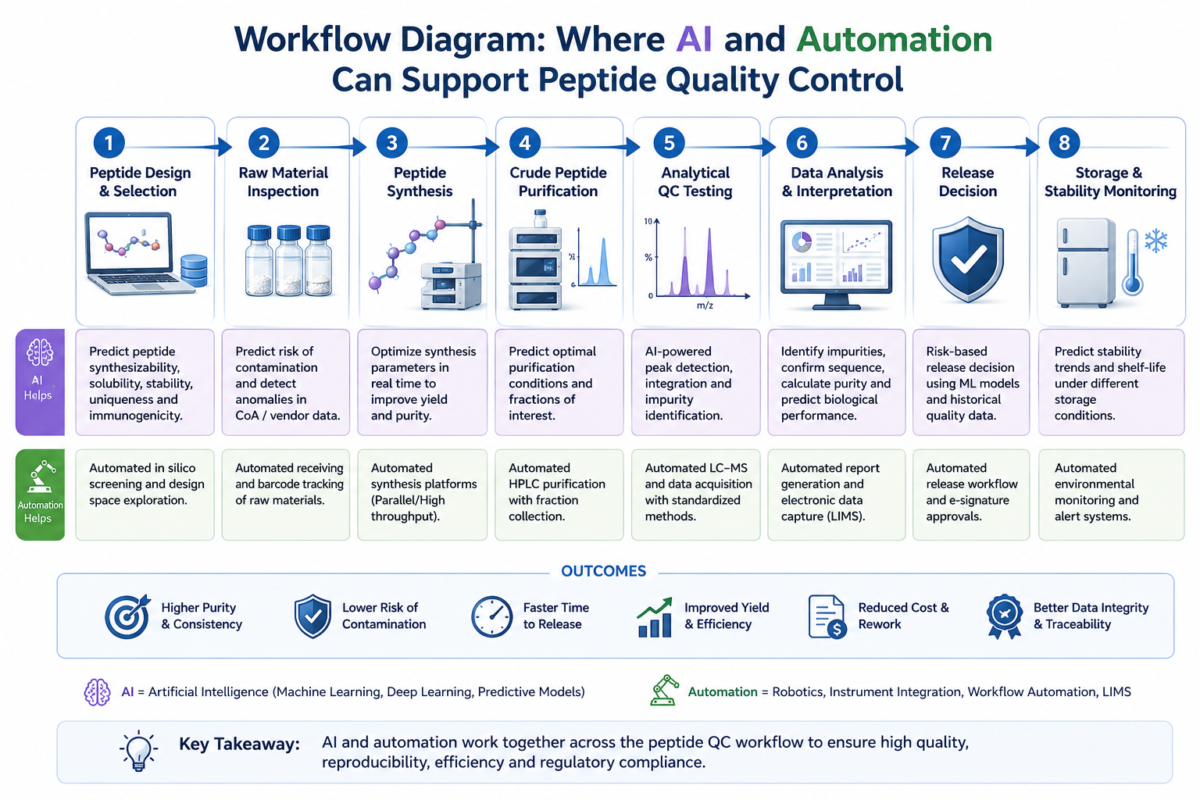
Figure 1: Workflow diagram showing where AI and automation can support peptide quality control.
Machine learning algorithms have also shown potential for identifying subtle analytical deviations that may be overlooked during manual review.[8] In some laboratory environments, AI systems are trained using historical chromatographic and mass spectrometry data to recognize abnormal peptide signatures. This capability may support earlier detection of synthesis inconsistencies or contamination risks.
Another area of interest involves predictive modeling. AI tools may evaluate process trends and instrument behavior to forecast potential analytical failures before they occur.[9] Predictive systems can strengthen peptide quality control by helping laboratories identify maintenance needs or calibration issues earlier in the workflow.
What Role Automation Plays in Peptide Quality Control Workflows
Automation already plays a major role in many quality control workflows involving peptide synthesis and analytical testing. Robotic liquid handling systems are widely used for sample preparation because they improve pipetting precision and reduce operator variability.[10] Automated sample preparation also decreases contamination risk during repetitive laboratory procedures. Instrument integration is another important aspect of automation.

Figure 2: Comparison chart of traditional vs AI-supported peptide quality control processes
Modern analytical platforms can automatically transfer chromatographic and mass spectrometry data into laboratory information management systems for centralized review and storage.[11] This integration improves traceability and simplifies documentation processes associated with peptide quality control.Automated environmental tracking systems have also improved laboratory monitoring practices. Sensors can continuously monitor temperature, humidity, and instrument performance to ensure stable analytical conditions.[12] Continuous laboratory monitoring may help laboratories detect deviations more rapidly than periodic manual inspections.
Automation can also improve workflow scalability. High-throughput peptide screening laboratories often process hundreds of analytical samples daily, making automated handling systems valuable for maintaining consistent processing speeds.[13] As analytical demand increases, automation may become increasingly integrated into routine peptide quality control operations.
Why Data Analysis Matters in Modern Peptide Quality Control
Data interpretation has become one of the most important aspects of peptide quality control because analytical instruments now generate large and highly detailed datasets. Chromatographic resolution, retention times, spectral fragmentation patterns, and impurity profiles must all be evaluated carefully to confirm peptide integrity.[14]
Advanced analytical software can help laboratories organize and compare historical testing data more effectively. Trend analysis tools may identify recurring deviations across batches, instruments, or synthesis conditions.[15] This type of evaluation strengthens quality control workflows by improving consistency between production cycles.
Centralized data systems also support audit readiness and traceability. Electronic laboratory systems can preserve analytical records, instrument parameters, and review histories in a searchable format.[16] These systems reduce the likelihood of transcription errors associated with paper-based documentation.
As AI and automation continue developing, laboratories may rely increasingly on integrated software ecosystems capable of combining analytical interpretation, reporting, and operational oversight within a unified quality platform.
AI and Automation vs Traditional Peptide Quality Control Methods
Traditional peptide quality control methods often rely heavily on manual review, handwritten documentation, and technician-dependent interpretation. Although these approaches remain effective, they may introduce variability between analysts or increase processing time during high-volume operations.[17]
AI and automation systems can reduce some of these limitations by standardizing repetitive procedures and improving analytical consistency. Automated review platforms may process chromatographic datasets more rapidly than manual review alone while maintaining predefined analytical criteria.[18] These technologies can also improve reporting speed and reduce administrative workload.
However, human oversight remains essential in peptide quality control. Experienced analytical scientists are still required to validate methods, investigate atypical findings, and interpret complex analytical outcomes. Current research supports combining automation with expert review rather than fully replacing laboratory personnel.[19]
Future Trends in Peptide Quality Control: What Comes Next?
Future trends in peptide quality control are expected to focus on greater digital integration, predictive analytics, and expanded automation capabilities. Laboratories are increasingly adopting interconnected analytical systems capable of sharing instrument data in real time across centralized platforms.[5]
Artificial intelligence may continue improving analytical interpretation by supporting impurity recognition, predictive maintenance, and automated data validation.[8] Advanced machine learning systems could also strengthen quality control workflows through earlier identification of process instability or instrument drift.
Cloud-based laboratory systems may further improve collaboration between manufacturing sites, analytical laboratories, and quality assurance teams.[16] These digital ecosystems can simplify data sharing while improving traceability across peptide production environments.
Although AI and automation technologies continue advancing, scientific oversight, validated analytical methods, and standardized testing procedures remain fundamental to reliable peptide quality control. Future developments will likely emphasize integration between automated systems and experienced analytical professionals rather than complete replacement of human expertise.
Frequently asked questions (FAQs) about AI and Automation in Peptide Quality Control
What is peptide quality control?
- Peptide quality control refers to the analytical processes used to verify the identity, purity, concentration, and safety of peptides during research and manufacturing workflows. Common peptide quality control methods include high-performance liquid chromatography (HPLC), mass spectrometry (MS), endotoxin testing, sterility testing, moisture analysis, and peptide sequencing verification. These procedures help laboratories confirm that a peptide batch meets predefined specifications before it is used in research applications.
How could AI support peptide quality control in the future?
- Artificial intelligence may support peptide quality control by helping laboratories analyze complex analytical data more efficiently. AI-assisted systems could potentially identify abnormal chromatographic peaks, detect instrument drift, recognize contamination patterns, and flag deviations from historical batch profiles. Machine learning models may also help improve predictive maintenance for analytical instruments and optimize workflow scheduling. In peptide research environments with high sample throughput, AI-driven software could help reduce manual review time while improving consistency in data interpretation.
What role does automation play in peptide quality control workflows?
- Automation helps standardize repetitive laboratory processes and reduce the risk of human error during peptide quality control. Automated systems are already used in sample preparation, liquid handling, HPLC injection sequences, environmental monitoring, endotoxin testing workflows, and electronic documentation management. By reducing manual intervention, automation can improve reproducibility, increase throughput, and support more consistent analytical performance across peptide batches.
What peptide quality control tasks are most likely to be automated?
Several peptide quality control tasks are strong candidates for automation due to their repetitive and data-intensive nature. These may include:
- Automated HPLC sample injection and chromatogram processing
- Robotic liquid handling for assay preparation
- Automated endotoxin testing workflows
- Electronic batch record generation and review
- Instrument calibration tracking and maintenance alerts
- AI-assisted impurity detection and peak integration
- Environmental monitoring data collection and trend analysis
As laboratory software and robotics continue to improve, additional quality control functions may become partially or fully automated in research and manufacturing environments.
What should labs consider before adopting AI and automation in quality control?
- Laboratories should evaluate several factors before implementing AI and automation systems in peptide quality control workflows. Important considerations include data integrity, software validation, instrument compatibility, regulatory expectations, cybersecurity, staff training requirements, and long-term maintenance costs. Labs should also confirm that automated systems can produce reproducible and auditable analytical results. While automation may improve efficiency, human oversight remains important for interpreting complex data, investigating deviations, and ensuring scientific accuracy.
References
- Swartz ME. UPLC™: An introduction and review. J Liq Chromatogr Relat Technol. 2005;28(7-8):1253-1263. Available at: UPLC™: An Introduction and Review
- Merrifield RB. Solid phase peptide synthesis. J Am Chem Soc. 1963;85(14):2149-2154. Available at: Solid Phase Peptide Synthesis
- Aebersold R, Mann M. Mass spectrometry-based proteomics. Nature. 2003;422(6928):198-207. Available at: Mass Spectrometry-Based Proteomics
- Glish GL, Vachet RW. The basics of mass spectrometry in the twenty-first century. Nat Rev Drug Discov. 2003;2(2):140-150. Available at: The Basics of Mass Spectrometry in the Twenty-First Century
- US Food and Drug Administration. Data Integrity and Compliance With Drug CGMP. FDA Guidance for Industry. 2018. Available at: FDA Data Integrity Guidance
- Elliott P, Peakman TC. The UK Biobank sample handling and storage protocol for the collection, processing and archiving of human blood and urine. Int J Epidemiol. 2008;37(2):234-244. Available at: UK Biobank Sample Handling and Storage Protocol
- Tarca AL, Carey VJ, Chen XW, Romero R, Drăghici S. Machine learning and its applications to biology. PLoS Comput Biol. 2007;3(6):e116. Available at: Machine Learning and Its Applications to Biology
- Camacho DM, Collins KM, Powers RK, Costello JC, Collins JJ. Next-generation machine learning for biological networks. Cell. 2018;173(7):1581-1592. Available at: Next-Generation Machine Learning for Biological Networks
- Lee J, Davari H, Singh J, Pandhare V. Industrial artificial intelligence for Industry 4.0-based manufacturing systems. Manuf Lett. 2018;18:20-23. Available at: Industrial Artificial Intelligence for Industry 4.0-Based Manufacturing Systems
- Sastri VR. Plastics in Medical Devices: Properties, Requirements, and Applications. Elsevier; 2014. Available at: Plastics in Medical Devices: Properties, Requirements, and Applications
- Taylor CF. Standards for reporting bioscience data: a forward look. Drug Discov Today. 2007;12(13-14):527-533. Available at: Standards for Reporting Bioscience Data
- Whyte W. Cleanroom Technology: Fundamentals of Design, Testing and Operation. 2nd ed. Wiley-Blackwell; 2010. Available at: Cleanroom Technology: Fundamentals of Design, Testing and Operation
- Gooding KM, Choudhary G. High-throughput analytical strategies in pharmaceutical analysis. J Pharm Biomed Anal. 2019;164:181-197. Available at: High-Throughput Analytical Strategies in Pharmaceutical Analysis
- Dong MW. Modern HPLC for Practicing Scientists. Wiley-Interscience; 2006. Available at: Modern HPLC for Practicing Scientists
- Brereton RG. Chemometrics for Pattern Recognition. Wiley; 2009. Available at: Chemometrics for Pattern Recognition
- Ludäscher B, et al. Scientific workflow management and the Kepler system. Concurr Comput Pract Exp. 2006;18(10):1039-1065. Available at: Scientific Workflow Management and the Kepler System
- Meyer VR. Practical High-Performance Liquid Chromatography. 5th ed. Wiley; 2010. Available at: Practical High-Performance Liquid Chromatography
- Topol EJ. High-performance medicine: the convergence of human and artificial intelligence. Nat Med. 2019;25(1):44-56. Available at: High-Performance Medicine: The Convergence of Human and Artificial Intelligence
- Jiang F, Jiang Y, Zhi H, et al. Artificial intelligence in healthcare: past, present and future. Stroke Vasc Neurol. 2017;2(4):230-243. Available at: Artificial Intelligence in Healthcare: Past, Present and Future
