Kinetic Chromogenic LAL for Real-Time LPS Peptide Monitoring
Author: Dr. Numan S. Date: April 2, 2026
What Is LPS Control?
Lipopolysaccharide (LPS) is a structurally conserved component of the outer membrane of Gram-negative bacteria and is widely recognized as a potent activator of innate immune responses through Toll-like receptor 4 signaling pathways [1]. Even trace quantities of LPS can induce cytokine release, making strict LPS monitoring essential in pharmaceutical and research environments involving peptides.
LPS control refers to both the prevention of endotoxin contamination and the verification that endotoxin levels remain within defined regulatory limits. These limits are typically expressed in endotoxin units (EU), and compliance is confirmed through validated analytical methods such as the Limulus Amebocyte Lysate assay. Effective LPS monitoring ensures that peptide materials meet quality standards and do not introduce unintended biological effects [1,2].
What Is Kinetic Chromogenic LAL?
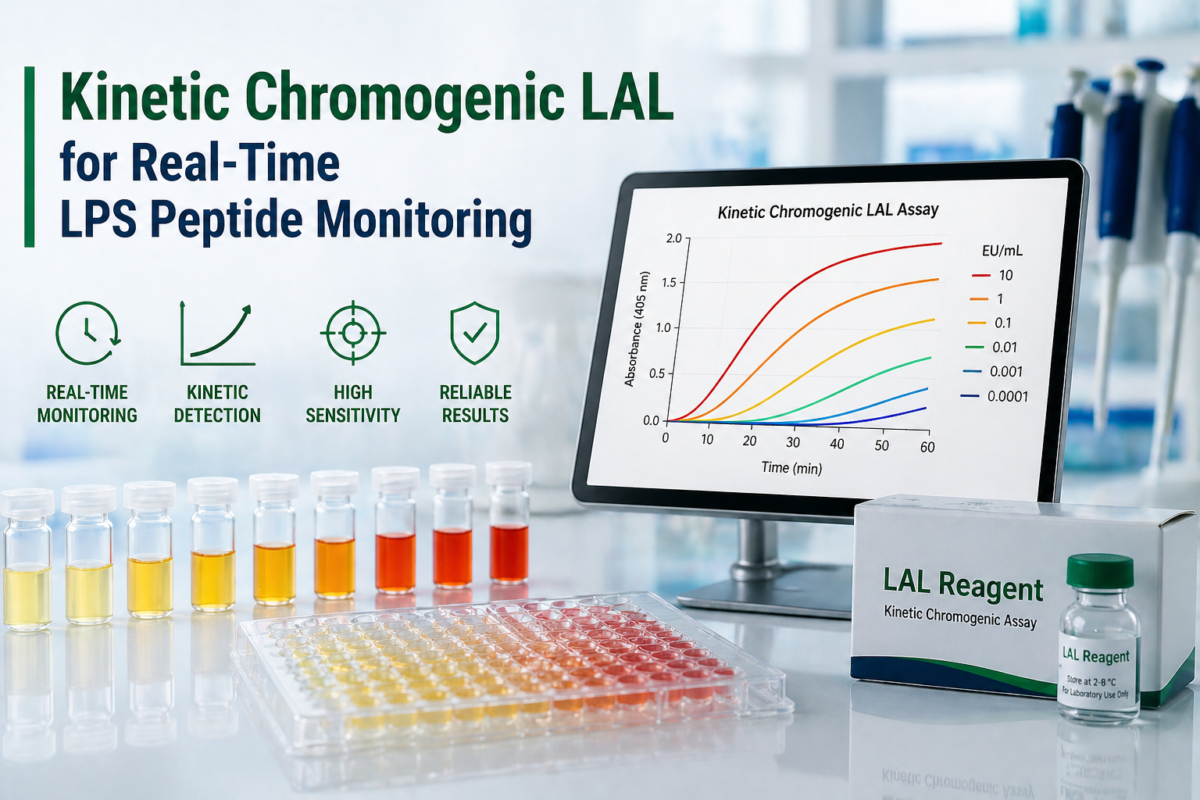
Kinetic chromogenic LAL is a quantitative variation of the LAL assay that measures endotoxin concentration based on the rate of chromogenic signal generation. In this method, endotoxin activates a cascade of enzymes derived from horseshoe crab amebocytes, ultimately leading to cleavage of a synthetic substrate that releases a colored compound, typically p-nitroaniline [3].
The defining feature of kinetic chromogenic LAL is its reliance on reaction kinetics rather than endpoint measurement. By continuously monitoring absorbance over time, this approach provides a dynamic assessment of endotoxin concentration. This makes kinetic chromogenic LAL particularly valuable for applications requiring precise and reproducible endotoxin detection in complex peptide matrices [2].
How Kinetic Chromogenic LAL Works for LPS Monitoring
The mechanism underlying kinetic chromogenic LAL begins with activation of factor C, a serine protease that specifically recognizes endotoxin. This activation triggers a proteolytic cascade involving factor B and a proclotting enzyme, ultimately resulting in cleavage of a chromogenic substrate [1,3]. In kinetic chromogenic LAL, absorbance is measured continuously at approximately 405 nm using a microplate reader.
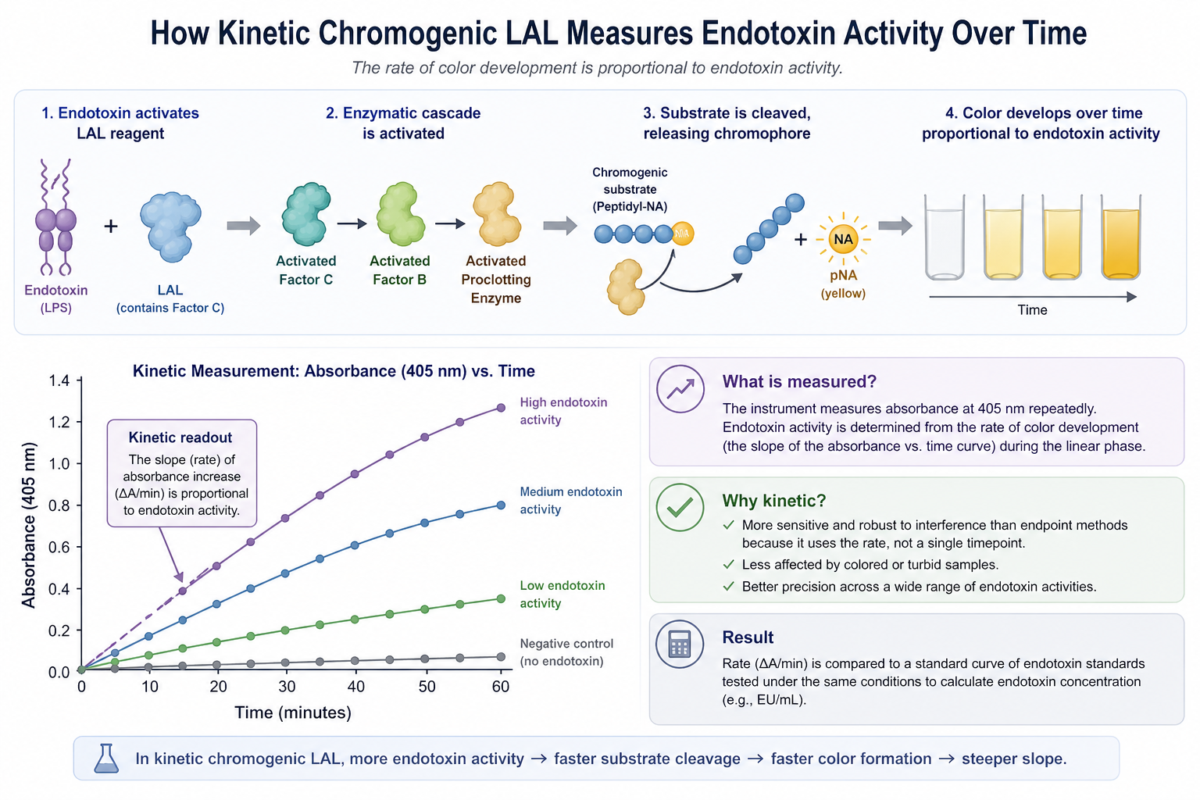
Figure 1: Kinetic chromogenic LAL process explained.
The time required to reach a predefined absorbance threshold is inversely proportional to endotoxin concentration. This enables accurate LPS monitoring across a wide dynamic range and allows detection of subtle differences in endotoxin levels [2]. Because the measurement is time-dependent, kinetic chromogenic LAL reduces variability associated with endpoint-only methods.
Why Real-Time LPS Monitoring Matters in Peptide Workflows
Real-time LPS monitoring is critical in peptide workflows because endotoxin contamination can occur at multiple stages, including synthesis, purification, and handling. Peptides are often processed in aqueous environments that support endotoxin persistence, and standard sterilization methods do not reliably eliminate LPS [1].
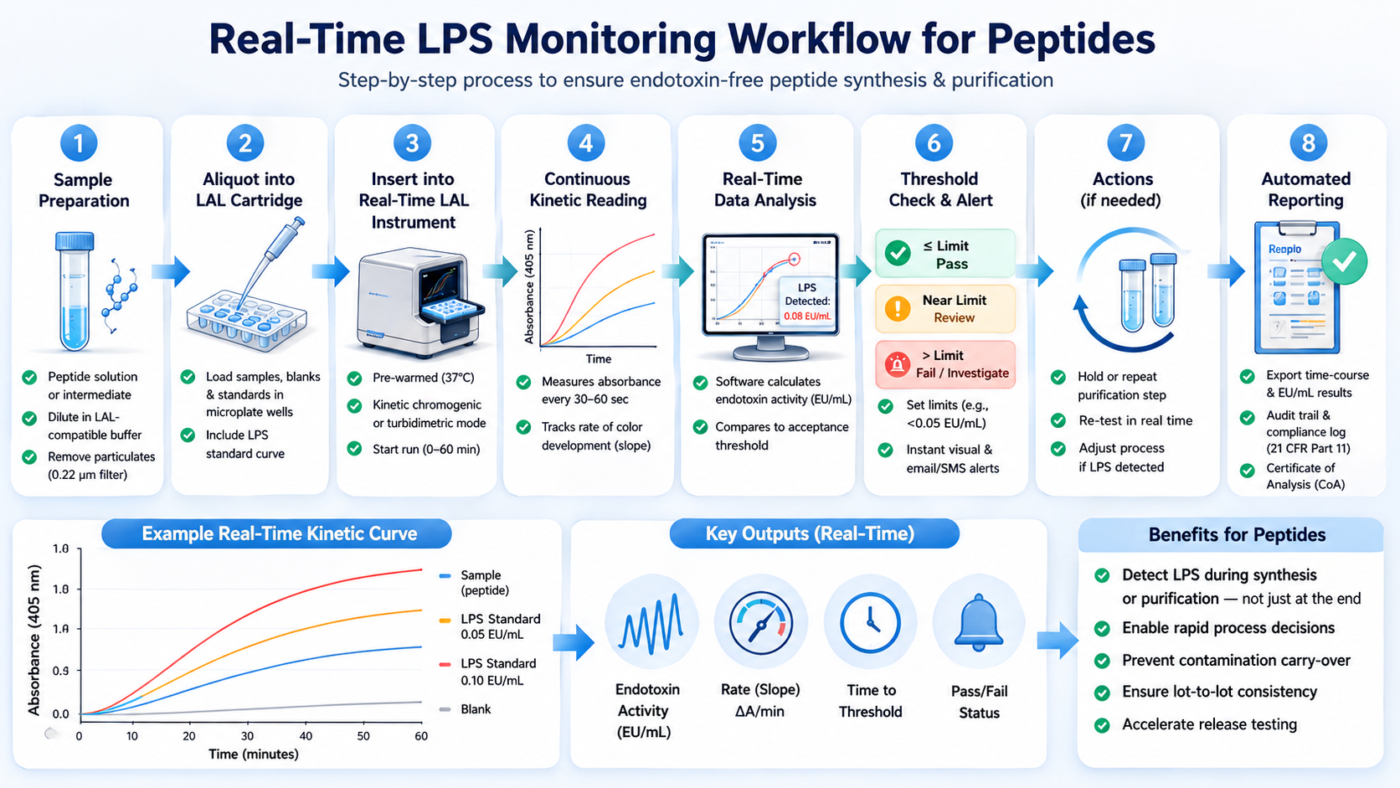
Figure 2: Step-by-step diagram of a real-time LPS monitoring workflow for peptides.
Studies have shown that endotoxin activity can change over time due to interactions with biological matrices, leading to decreased detectability in delayed measurements [4]. Without real-time monitoring, these changes can result in inaccurate endotoxin detection. Kinetic chromogenic LAL addresses this limitation by capturing the kinetics of endotoxin activity as it occurs, providing a more reliable assessment of contamination risk during peptide testing.
How Peptide Samples Can Affect LPS Monitoring Results
Peptide samples can significantly influence the accuracy of LPS monitoring due to matrix effects. Many peptides, particularly cationic sequences, interact with endotoxin molecules by binding to the negatively charged lipid A region. This interaction can reduce the accessibility of endotoxin to the LAL assay reagents [1].
This effect is commonly referred to as low endotoxin recovery (LER). In such cases, endotoxin may be present but not fully detectable, resulting in falsely low readings. Importantly, research demonstrates that masked endotoxin can retain biological activity despite reduced detection by LAL-based methods [5]. These findings highlight the importance of validating kinetic chromogenic LAL specifically for peptide testing applications.
What To Know About Sensitivity and Detection Range
Kinetic chromogenic LAL is known for its high sensitivity and broad detection range, typically spanning from approximately 0.005 to 50 EU/mL depending on assay configuration [3]. This sensitivity makes it suitable for detecting low levels of endotoxin in peptide samples.
However, assay performance is influenced by sample composition. Interfering substances such as salts, surfactants, and solvents can inhibit or enhance the enzymatic cascade, affecting accuracy. To address these challenges, validation procedures include spike recovery experiments and dilution linearity assessments, ensuring that LPS monitoring results reflect true endotoxin levels [1,2].
Kinetic Chromogenic LAL vs Gel Clot and Turbidimetric Methods
The LAL assay exists in three primary formats: gel clot, turbidimetric, and chromogenic. Gel clot methods provide qualitative results based on clot formation, while turbidimetric assays measure changes in turbidity as the reaction progresses [1].
Kinetic chromogenic LAL differs by measuring color development rather than turbidity. This distinction offers several advantages, including improved sensitivity and compatibility with samples that may interfere with turbidity measurements. Additionally, kinetic chromogenic LAL provides continuous data, enabling more precise endotoxin detection compared to endpoint-based approaches [2]. These features make it a preferred method for advanced LPS monitoring applications.
Kinetic Chromogenic LAL vs Gel Clot and Turbidimetric Methods
The LAL assay exists in three primary formats: gel clot, turbidimetric, and chromogenic. Gel clot methods provide qualitative results based on clot formation, while turbidimetric assays measure changes in turbidity as the reaction progresses [1].
Kinetic chromogenic LAL differs by measuring color development rather than turbidity. This distinction offers several advantages, including improved sensitivity and compatibility with samples that may interfere with turbidity measurements. Additionally, kinetic chromogenic LAL provides continuous data, enabling more precise endotoxin detection compared to endpoint-based approaches [2]. These features make it a preferred method for advanced LPS monitoring applications.
Frequently asked questions (FAQs) about Kinetic Chromogenic LAL in Peptide Testing
What is kinetic chromogenic LAL?
- Kinetic chromogenic Limulus Amebocyte Lysate (LAL) is a quantitative endotoxin detection assay that measures lipopolysaccharide (LPS) activity based on a time-dependent color reaction. When endotoxin is present, it activates a proteolytic enzyme cascade derived from horseshoe crab blood, which cleaves a synthetic chromogenic substrate. This releases a colored compound, and the rate of color development—measured spectrophotometrically over time—is directly proportional to the endotoxin concentration in the sample.
How does kinetic chromogenic LAL support real-time LPS monitoring in peptides?
- Unlike endpoint assays, kinetic chromogenic LAL continuously measures absorbance changes during the reaction, allowing for real-time tracking of endotoxin activity. In peptide workflows, this enables researchers to detect LPS levels dynamically as the reaction progresses, improving sensitivity and quantification accuracy. The assay generates a standard curve based on reaction time (onset time), which allows precise interpolation of endotoxin concentrations in unknown peptide samples.
Why is real-time LPS monitoring important in peptide workflows?
- Real-time monitoring enhances both sensitivity and process control. Peptides—especially synthetic or purified compounds—can be susceptible to trace endotoxin contamination that may not be detected with less sensitive methods. Continuous kinetic measurement reduces variability, improves reproducibility, and allows early detection of contamination during critical stages such as purification, reconstitution, or formulation. This is particularly important in research settings where endotoxin presence can interfere with biological assays or skew experimental outcomes.
How does kinetic chromogenic LAL compare to other LAL methods?
- Kinetic chromogenic LAL offers several advantages over traditional gel-clot and endpoint chromogenic assays. Compared to the gel-clot method, it provides quantitative rather than qualitative results and significantly higher sensitivity. Compared to endpoint chromogenic assays, it reduces operator variability by eliminating reliance on a single time-point measurement. Additionally, kinetic methods allow for automated data collection and analysis, making them well-suited for high-throughput peptide testing environments. However, it may require more specialized instrumentation, such as a microplate reader capable of kinetic measurements.
What should researchers know before using kinetic chromogenic LAL for peptide testing?
- Researchers should be aware of potential assay interferences from peptide matrices, including pH extremes, solvents, or excipients that may inhibit or enhance the LAL reaction. Proper validation—such as inhibition/enhancement testing (spike recovery)—is essential to confirm assay reliability for a given peptide formulation. Additionally, strict endotoxin-free lab practices, including the use of depyrogenated glassware and certified LAL-grade reagents, are critical. Calibration with validated endotoxin standards and adherence to regulatory or pharmacopeial guidelines (e.g., USP <85>) further ensure accurate and reproducible results.
