LPS Control Strategies in the Peptide Production Lifecycle
Author: Dr. Numan S. Date: March 26, 2026

Learn how LPS control supports cleaner peptide production by reducing endotoxin risk across sourcing, handling, purification, testing, and storage stages. Lipopolysaccharide (LPS), commonly referred to as endotoxin, is a structural component of Gram-negative bacterial outer membranes. Its biologically active lipid A region is responsible for strong immune responses even at low concentrations [1].
LPS control refers to the systematic prevention, monitoring, and reduction of endotoxin contamination throughout manufacturing workflows. Effective lps control focuses on minimizing introduction sources and verifying removal at critical stages where downstream correction becomes difficult.
Why LPS Control Matters in Peptide Production
In peptide production, endotoxin contamination presents a critical quality risk, particularly for applications requiring high purity and biological reliability. Even trace endotoxin levels can interfere with experimental outcomes or trigger inflammatory responses in biological systems [2].
Regulatory frameworks define strict endotoxin thresholds, often based on dose-dependent exposure limits such as 5 EU/kg for most routes [2]. These limits emphasize why lps control must be integrated across the entire production lifecycle rather than addressed only at final testing.
How LPS Can Enter the Peptide Production Lifecycle
Endotoxin contamination can occur at multiple stages of peptide production. Common sources include process water, raw materials, equipment surfaces, and environmental exposure during handling [2,6]. Notably, endotoxins are highly stable molecules that persist even after microbial death. Standard sterilization methods may eliminate bacteria but do not remove endotoxin contamination already present, making early-stage lps control essential.
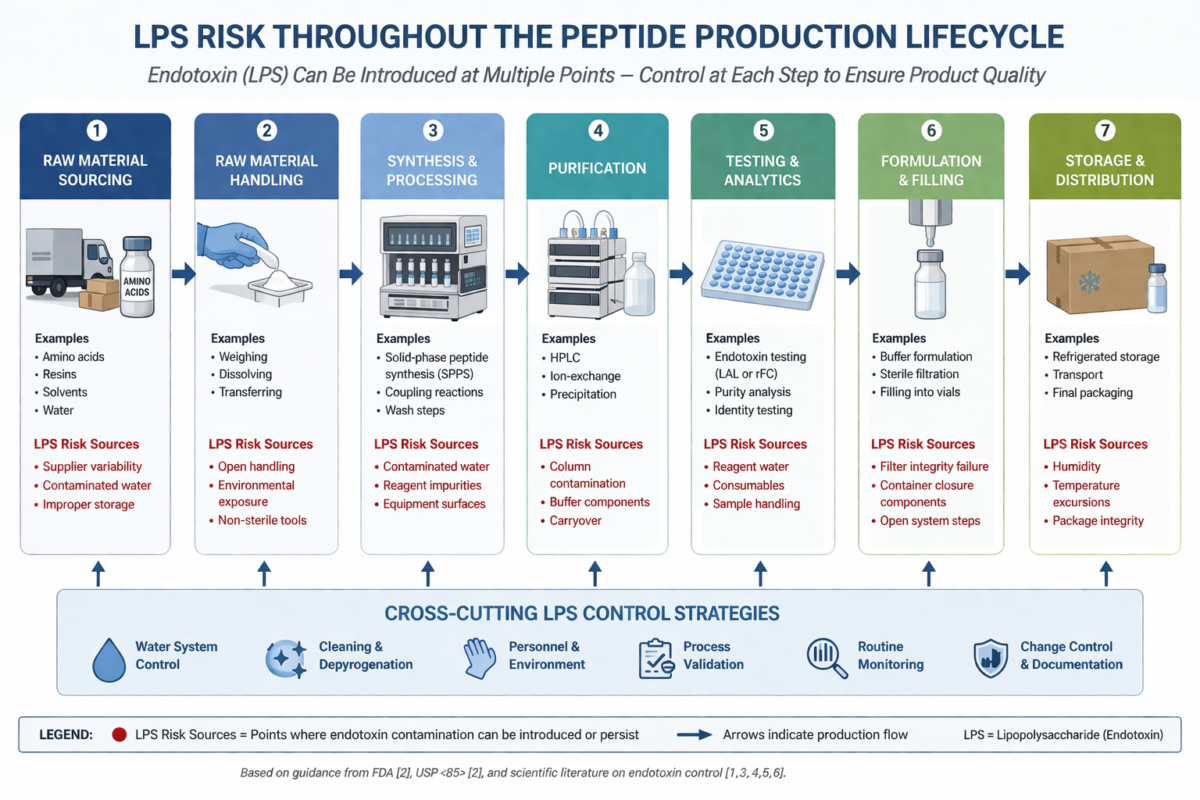
Figure 1: Lifecycle diagram showing where LPS risk can appear during peptide production
Raw material control is a foundational aspect of lps control. Supplier qualification and verification of endotoxin levels in incoming materials reduce the risk of introducing contaminants early in peptide production [6]. Controlled storage conditions, including temperature and humidity management, help prevent microbial proliferation that can generate endotoxins. Segregation of approved and quarantined materials further supports consistent control practices.
Water systems represent one of the most significant contributors to endotoxin contamination. High-temperature circulating systems (>75°C) are widely recognized for limiting microbial growth and endotoxin formation [2]. Process controls such as defined hold times, closed-system transfers, and controlled environmental exposure reduce the likelihood of contamination during intermediate stages. These measures strengthen overall lps control by minimizing variability.
Endotoxins exhibit strong surface adhesion and resistance to conventional sterilization. Effective lps control requires validated cleaning methods, including alkaline washes and oxidizing agents followed by endotoxin-free rinsing [2].Dry heat depyrogenation remains a gold-standard approach for endotoxin destruction. Studies demonstrate that exposure to temperatures around 250°C can significantly reduce endotoxin levels, supporting its use in equipment preparation [4].
LPS Control During Peptide Purification and Processing
The purification process plays a critical role in endotoxin removal. However, endotoxins can associate with peptides through hydrophobic and electrostatic interactions, complicating separation [3].Chromatographic techniques, including ion-exchange systems, as well as detergent-based phase separation methods, have been shown to reduce endotoxin contamination when optimized appropriately [3,6]. Integrating these strategies into the purification process enhances lps control without compromising yield.
Endotoxin testing provides essential verification of lps control effectiveness. The Limulus amebocyte lysate (LAL) assay remains a widely used method, with gel-clot, chromogenic, and turbidimetric variations available [2]. Recombinant factor C (rFC) assays offer an alternative with comparable sensitivity and reliability. Comparative studies demonstrate equivalent performance between LAL and rFC methods when properly validated [5]. These tools ensure consistent monitoring throughout peptide production.
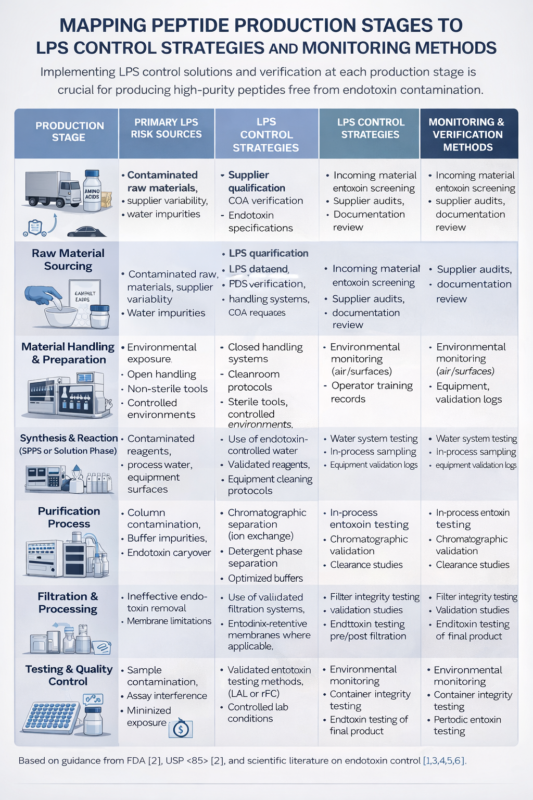
Figure 2: Table mapping production stages to LPS control strategies and monitoring methods
Packaging components such as vials and stoppers can introduce endotoxin contamination if not properly treated. Washing with endotoxin-free water and minimizing post-clean handling are essential practices [2]. Storage conditions must prevent moisture accumulation and microbial growth. Controlled environments and sealed systems help maintain the integrity of materials and support ongoing lps control.
Common Challenges in Maintaining LPS Control
LPS control is inherently challenging due to the unique physicochemical properties of endotoxins. Structurally, lipopolysaccharides are amphipathic molecules composed of a hydrophobic lipid A domain and a hydrophilic polysaccharide chain. This dual nature promotes aggregation in aqueous environments and facilitates strong interactions with proteins, peptides, and surfaces, significantly complicating removal during downstream processing [1,3]. In peptide systems specifically, hydrophobic interactions and electrostatic binding can lead to endotoxin co-purification, reducing the efficiency of conventional separation techniques.
In addition to removal challenges, endotoxin contamination is difficult to detect accurately under certain conditions. Analytical limitations arise from matrix interference in endotoxin testing assays, particularly those based on Limulus amebocyte lysate (LAL) methodologies. Variations in pH, ionic strength, and the presence of surfactants or organic solvents can inhibit or enhance assay responses, leading to false-negative or false-positive results [2]. Adsorption of endotoxins to container surfaces or sample components can further obscure true concentrations, necessitating rigorous method suitability testing and spike recovery validation to ensure reliable measurements.

Figure 3: Table mapping production stages to LPS control strategies and monitoring methods
Another complicating factor in lps control is the persistence of endotoxins despite microbial inactivation. Because endotoxins are not living entities, standard sterilization techniques—such as filtration or autoclaving—may eliminate viable bacteria without removing residual endotoxin contamination. This disconnect underscores the importance of distinguishing between sterility assurance and endotoxin control, as both require separate validation strategies within peptide production environments.
Effective lps control therefore demands a comprehensive, lifecycle-based approach that integrates prevention, monitoring, and validated removal strategies. Identifying high-risk control points such as water systems, raw material interfaces, and open handling steps enables targeted interventions that reduce the likelihood of endotoxin introduction [6]. Preventive measures, including closed-system processing, validated depyrogenation procedures, and strict environmental controls, are essential for minimizing contamination risk at its source.
Equally important is the implementation of continuous monitoring systems and data-driven process control. Routine endotoxin testing at multiple stages, combined with environmental monitoring and water system surveillance, allows for early detection of contamination trends. Trend analysis and statistical process control can reveal subtle deviations before they result in out-of-specification outcomes, strengthening overall process robustness.
Best Practices for a More Reliable LPS Control Strategy
A robust lps control strategy requires a multi-layered, risk-based approach that integrates prevention, removal, and verification across the entire manufacturing workflow. Rather than relying on downstream correction, effective programs prioritize upstream controls that limit endotoxin contamination before it becomes difficult to remove [2,6].
First, raw material qualification should include supplier audits, endotoxin specifications, and traceable documentation. Water systems—one of the most common sources of endotoxin contamination—must be continuously monitored, maintained at controlled temperatures, and validated for microbial and endotoxin limits [2].Second, validated cleaning and depyrogenation protocols are essential. Dry heat depyrogenation for heat-stable components and chemically validated cleaning procedures for process equipment should be routinely verified through endotoxin challenge studies [4]. These practices ensure that equipment does not serve as a persistent reservoir for contamination.
Third, the purification process should be designed with built-in endotoxin clearance mechanisms. Chromatographic techniques, phase separation methods, and optimized buffer conditions can significantly reduce endotoxin contamination when selected based on molecular interactions between peptides and LPS [3,6]. Fourth, analytical verification through endotoxin testing must be method-specific and validated for the product matrix. Routine testing at multiple production stages—not just final release—enables early detection of contamination trends and supports continuous process improvement [5].
Finally, a comprehensive lps control program includes environmental monitoring, personnel training, and strict handling protocols. Controlled environments, minimized open processing steps, and proper storage and packaging conditions collectively reduce the risk of reintroduction after purification.By integrating these best practices, manufacturers can establish a resilient lps control framework that supports consistent peptide production quality, minimizes endotoxin contamination risk, and aligns with regulatory expectations for high-purity biological materials.
Frequently asked questions (FAQs) about LPS Control Strategies in the Peptide Production Lifecycle
What is LPS control?
- LPS control refers to the systematic prevention, reduction, and monitoring of lipopolysaccharide (LPS) contamination throughout peptide production workflows. LPS, a component of the outer membrane of Gram-negative bacteria, is a potent endotoxin that can interfere with analytical results and compromise research integrity. Effective LPS control integrates facility design, material selection, process controls, and validated testing methods to ensure endotoxin levels remain within acceptable limits for research applications.
Why does LPS control matter throughout the peptide production lifecycle?
- LPS control is critical because contamination can occur at any stage of production and is difficult to remove once introduced. Even trace levels of endotoxin can interfere with biological assays, alter cellular responses, and compromise experimental reproducibility. Maintaining strict LPS control ensures that peptide samples remain reliable for analytical characterization and downstream research applications, preserving data integrity across studies.
How can LPS contamination enter peptide production workflows?
- LPS contamination can enter peptide workflows through multiple pathways, often originating from environmental exposure or material inputs. Water systems that are not endotoxin-free represent a major risk, as do contaminated raw materials such as amino acids or buffers. In addition, improperly cleaned equipment, reusable chromatography components, and laboratory surfaces can harbor endotoxins. Human handling and airborne particulates in poorly controlled environments further increase the likelihood of contamination during synthesis, purification, or filling processes.
What strategies help reduce endotoxin risk during production?
- Reducing endotoxin risk requires a proactive and controlled production environment. This includes the use of endotoxin-free water, strict cleanroom protocols, and validated depyrogenation methods such as high-temperature dry heat treatment for glassware. Equipment must be thoroughly cleaned and verified between uses, and the use of single-use consumables can further minimize contamination risk. Additionally, controlling raw material quality through supplier qualification and limiting manual handling through proper aseptic technique are essential components of an effective endotoxin prevention strategy.
How is LPS control monitored across different production stages?
- LPS control is monitored through a combination of routine testing and process validation at key stages of production. Raw materials are often screened prior to use, while in-process testing may include monitoring water systems and intermediate solutions. Final product testing confirms that endotoxin levels meet required specifications before release. Analytical methods such as the Limulus Amebocyte Lysate (LAL) assay, recombinant Factor C (rFC), and Monocyte Activation Test (MAT) are commonly used, with results reported in endotoxin units per milliliter (EU/mL). Continuous environmental monitoring of cleanroom conditions also supports consistent LPS control throughout the production lifecycle.
References
- Rietschel ET, Kirikae T, Schade FU, et al. Bacterial endotoxin: molecular relationships of structure to activity and function. FASEB J. 1994;8(2):217-225.
- U.S. Food and Drug Administration. Bacterial Endotoxins/Pyrogens. 2014.
- Petsch D, Anspach FB. Endotoxin removal from protein solutions. J Biotechnol. 2000;76(2-3):97-119.
- Nakata T. Destruction of typical endotoxins by dry heat as determined using LAL assay and pyrogen assay. J Parenter Sci Technol. 1993;47(5):258-264.
- Piehler M, Roeder J, Fath S, et al. Comparison of LAL and rFC assays. Microorganisms. 2020;8(3):418.
- Schneier M, Razdan S, Miller AM, et al. Current technologies for endotoxin detection and removal. Biotechnol Bioeng. 2020;117(8):2588-2609.
