LPS Detection Sensitivity and EU/mL Specifications
Author: Dr. Numan S. Date: March 11, 2026

Endotoxin contamination can alter experimental outcomes because lipopolysaccharides activate innate immune pathways such as TLR4 signaling [2,3]. Accurate interpretation requires understanding lps detection sensitivity and not relying on simple positive or negative outcomes [1,5].
What Is LPS Detection Sensitivity?
LPS detection sensitivity refers to the lowest endotoxin concentration that an assay can reliably detect or quantify, typically expressed in EU/mL. This defines the lower boundary of analytical performance and determines whether trace contamination can be identified [5].
Endotoxin detection sensitivity must be validated within the actual sample matrix. Spike recovery experiments, typically requiring 50–200% recovery, confirm that assay performance remains reliable and reflects true endotoxin assay sensitivity rather than matrix interference [1,5].
Understanding Lipopolysaccharides (LPS)
Lipopolysaccharides are structural components of Gram-negative bacterial outer membranes, composed of lipid A, a core oligosaccharide, and an O-antigen [2,3]. Lipid A is responsible for most biological activity and inflammatory signaling, interacting with host receptors such as Toll-like receptor 4 (TLR4) to initiate cytokine production and immune activation [2,3].
LPS molecules aggregate in aqueous environments, forming supramolecular structures that complicate lipopolysaccharide detection. These structural changes directly influence LPS testing sensitivity and contribute to variability in endotoxin detection methods [2,4].
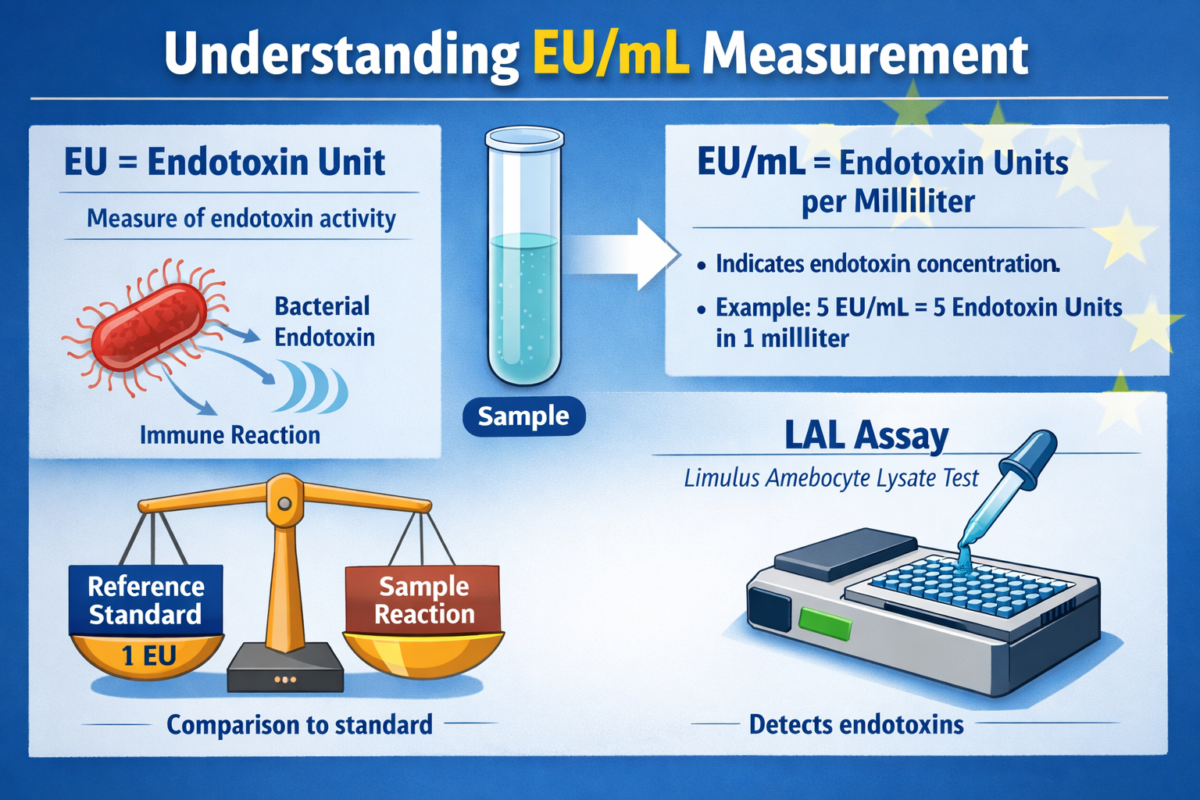
Figure 1: Understanding EU/mL measurement process
The amphipathic nature of LPS drives the formation of micelles, vesicles, or larger aggregates depending on concentration, ionic strength, and the presence of proteins or surfactants. These structural states can shield lipid A from interaction with assay reagents, reducing apparent endotoxin detection sensitivity and contributing to phenomena such as low endotoxin recovery (LER) [4].
Additionally, heterogeneity in LPS structure across bacterial species introduces variability in biological potency and assay response. Differences in acylation patterns of lipid A or variations in O-antigen length can alter both immune activation and assay detectability, further complicating LPS quantification methods [2]. This structural diversity underscores why endotoxin assay sensitivity must always be interpreted within the context of the specific sample matrix and experimental conditions.
What EU/mL Means in Endotoxin Testing
EU/mL endotoxin measurement represents biological activity rather than mass concentration. One endotoxin unit (EU) is standardized against reference endotoxin preparations and is considered equivalent to one international unit (IU) [5].
Because EU reflects biological potency, conversions between EU and mass (e.g., ng) vary depending on the reference standard. For this reason, EU/mL endotoxin measurement is preferred for comparing endotoxin testing limits across laboratories [2,5].
How LPS Detection Sensitivity Is Measured
LPS detection sensitivity is determined through calibration curves or labeled sensitivities (λ) depending on the assay format. Quantitative assays rely on standard curves, while gel-clot methods define sensitivity based on the lowest concentration producing a clot [5].
LAL assays can achieve high endotoxin detection sensitivity, with kinetic chromogenic formats reporting detection limits as low as 0.005 EU/mL under validated conditions [1]. However, matrix effects can reduce effective endotoxin assay sensitivity if not properly controlled.

Figure 2: LPS detection methods comparison table
In quantitative formats, calibration curves are generated using serial dilutions of a reference standard endotoxin, allowing EU/mL endotoxin measurement across a defined dynamic range. The lower limit of detection and lower limit of quantification are established based on signal-to-noise ratios and statistical validation, ensuring that reported values reflect true endotoxin presence rather than background variability [1,5].
Method suitability testing is a critical component of determining endotoxin detection sensitivity. This involves spiking known amounts of endotoxin into the sample matrix and verifying acceptable recovery, typically within 50–200%. Failure to meet these criteria indicates interference, which may require dilution, pH adjustment, or other pretreatment strategies to restore reliable LPS testing sensitivity [5].
Additionally, maximum valid dilution (MVD) calculations are used to ensure that sample dilution does not exceed assay capability while still meeting endotoxin testing limits. This balance is essential for maintaining endotoxin assay sensitivity in complex matrices, particularly in peptide formulations where binding interactions or excipients may suppress detectable signal [3,5].
Common Methods for Measuring LPS Detection Sensitivity
The most widely used endotoxin detection methods include Limulus amebocyte lysate (LAL) assays in gel-clot, turbidimetric, and chromogenic formats. These rely on activation of Factor C in horseshoe crab lysate [1,5].
Recombinant Factor C (rFC) assays provide an alternative with reduced interference from β-glucans. Combining LPS quantification methods such as LAL and rFC can improve reliability in complex matrices and enhance LPS testing sensitivity [1].
Matrix interference is a primary factor affecting endotoxin detection sensitivity. Substances such as detergents, salts, and organic solvents can inhibit or enhance assay reactions, leading to inaccurate results [3].
Low endotoxin recovery (LER) occurs when LPS becomes masked by formulation components, reducing apparent endotoxin detection sensitivity. This phenomenon is well documented and can significantly distort lipopolysaccharide detection outcomes [4].
Interpreting EU/mL Specifications in Laboratory Reports
EU/mL values must be interpreted alongside assay validation parameters, including recovery controls and dilution factors. Without these controls, reported values may not reflect true endotoxin levels [5].
Endotoxin testing limits are often defined based on application-specific thresholds. For example, parenteral products are typically evaluated against dose-based limits such as 5 EU/kg/hour, emphasizing the importance of context in EU/mL endotoxin measurement [3,5].
Peptide samples present unique challenges for lps detection sensitivity due to potential contamination during synthesis and handling. Additionally, peptides may bind LPS, reducing detectable signal and affecting endotoxin detection sensitivity [3].
This interaction can artificially improve apparent LPS testing sensitivity by masking endotoxin. Therefore, peptide workflows require validated spike recovery and, when necessary, orthogonal endotoxin detection methods to confirm results [1,3,5].
Best Practices for Maintaining High Detection Sensitivity
Maintaining high endotoxin detection sensitivity requires strict contamination control. Depyrogenation of materials, use of endotoxin-free consumables, and controlled environments reduce baseline endotoxin levels [5].
Analytical practices such as appropriate dilution, use of positive controls, and validation of pretreatment steps are essential. These measures ensure that LPS quantification methods accurately reflect true endotoxin levels rather than assay artifacts [3,5].
A common misconception is that kit sensitivity equals real-world performance. In practice, lps detection sensitivity depends heavily on sample composition and matrix effects.
Another misunderstanding is that “non-detectable” endotoxin implies absence. In reality, results below detection thresholds may still contain endotoxin below endotoxin testing limits, emphasizing the importance of endotoxin assay sensitivity.
Why LPS Detection Sensitivity Matters for Research Integrity
Endotoxin contamination can significantly alter biological outcomes, including cytokine release and cellular responses, even at low concentrations [2,3]. This makes accurate lipopolysaccharide detection critical for reproducibility.
Reliable lps detection sensitivity ensures that experimental results are not confounded by hidden endotoxin effects. Proper validation and reporting strengthen confidence in endotoxin detection methods and research conclusions [1,5].
Frequently asked questions (FAQs) about LPS Detection Sensitivity and Endotoxin Measurement
What does LPS detection sensitivity mean in laboratory testing?
- LPS detection sensitivity refers to the lowest concentration of lipopolysaccharide (endotoxin) that an assay can reliably detect and quantify. It is typically defined by the assay’s limit of detection (LOD) and limit of quantification (LOQ). High-sensitivity assays can detect very low endotoxin levels (e.g., ≤0.01 EU/mL), which is critical in pharmaceutical and peptide research where even trace endotoxin contamination may impact experimental outcomes or safety assessments. Sensitivity varies by method, reagent quality, and sample matrix.
How is EU/mL used to measure endotoxin levels?
- Endotoxin levels are expressed in endotoxin units per milliliter (EU/mL), a standardized unit based on the biological activity of a reference endotoxin preparation. One EU roughly corresponds to a specific amount of endotoxin that produces a defined response in the Limulus Amebocyte Lysate (LAL) assay. Reporting results in EU/mL allows for consistent comparison across assays and laboratories, even when different endotoxin sources or detection platforms are used.
What factors influence the sensitivity of LPS detection assays?
- Several variables affect assay sensitivity, including the detection method (e.g., LAL, recombinant Factor C), reagent quality, instrument calibration, and operator technique. Sample-related factors are also critical—these include pH, ionic strength, presence of surfactants, and peptide-specific interactions that may enhance or inhibit detection. Endotoxin masking, where LPS binds to peptides or excipients, can reduce apparent sensitivity and lead to falsely low readings if not properly addressed.
What testing methods detect lipopolysaccharides?
- The most widely used methods include Limulus Amebocyte Lysate (LAL) assays (gel clot, turbidimetric, and chromogenic formats), recombinant Factor C (rFC) assays, and the Monocyte Activation Test (MAT). LAL and rFC assays directly detect endotoxin via activation of clotting or fluorescence pathways, while MAT measures cytokine release from human immune cells in response to pyrogens, including LPS. Each method differs in sensitivity, specificity, and susceptibility to interference.
How should laboratories interpret endotoxin detection thresholds?
- Endotoxin thresholds should be interpreted in the context of the intended application, regulatory guidance, and assay limitations. For pharmaceutical products, thresholds are often defined by standards such as USP <85>, which specify allowable endotoxin limits based on dosage and route of administration. In research settings, lower thresholds may be applied to minimize biological interference. Laboratories should consider assay sensitivity, potential masking effects, and validation data when determining whether a sample meets acceptable endotoxin criteria.
References
- Piehler M, Roeder R, Blessing S, Reich J. Comparison of LAL and rFC assays—participation in a proficiency test program between 2014 and 2019. Microorganisms. 2020;8(3):418. doi:10.3390/microorganisms8030418
- Kimble A, Hauschild J, McDonnell G. Affinity and inactivation of bacterial endotoxins for medical device materials. Biomed Instrum Technol. 2023;57(4):153-162. doi:10.2345/0899-8205-57.4.153
- Harm S, Schildböck C, Cont D, et al. Heparin enables the reliable detection of endotoxin in human serum samples using the Limulus amebocyte lysate assay. Sci Rep. 2024;14:2410. doi:10.1038/s41598-024-52735-8
- Gorman A, Golovanov AP. Lipopolysaccharide structure and the phenomenon of low endotoxin recovery. Eur J Pharm Biopharm. 2022;180:289-307. doi:10.1016/j.ejpb.2022.10.006
- European Medicines Agency. ICH Q4B Annex 14: Bacterial endotoxins tests. Published 2010.
