LPS Removal Efficiency: Evaluating Different Purification Resins
Author: Dr. Numan S. Date: March 19, 2026

Learn how LPS detection sensitivity is measured in EU/mL, why it matters for endotoxin testing, and how labs interpret sensitivity thresholds in peptides.
Endotoxin contamination is a well-documented challenge in biologics and peptide workflows, particularly when materials are exposed to Gram-negative bacterial components such as lipopolysaccharide (LPS). LPS removal is therefore a critical downstream step to ensure accurate analytical results and reliable experimental outcomes [1].
In laboratory practice, endotoxin levels are quantified in endotoxin units (EU), typically expressed as EU/mL. These values directly inform LPS removal efficiency, making assay sensitivity and reporting format essential for interpreting purification outcomes [2].
Because LPS can persist through multiple processing steps, LPS removal strategies especially those involving purification resins are widely used to reduce endotoxin burden without compromising product integrity [3].
What Is LPS Removal?
LPS removal refers to the reduction of measurable endotoxin levels from a sample, typically expressed as EU/mL or EU per mass of product. The biologically active component, lipid A, is responsible for the majority of endotoxin activity [1].
Effective LPS removal requires eliminating or reducing endotoxin contamination to levels compatible with downstream applications while maintaining structural integrity of the target molecule [2].
LPS molecules are amphipathic and can form aggregates or bind to proteins, allowing them to persist through filtration and chromatography processes [3]. This makes LPS removal a necessary dedicated step rather than a passive outcome of purification. Additionally, endotoxin detection assays may underestimate endotoxin levels due to masking effects, where LPS binds biomolecules and reduces detectable activity, complicating endotoxin removal validation [4].
Purification resins support LPS removal by exploiting electrostatic and hydrophobic interactions. LPS carries a net negative charge due to phosphate groups, enabling binding to positively charged ligands on chromatographic media [5].
Affinity-based purification resins, including polymyxin B or polycationic ligands, can selectively bind lipid A, facilitating endotoxin removal from solution while allowing the target molecule to remain unbound [6].
What Affects LPS Removal Efficiency?
LPS removal efficiency depends on multiple variables, including pH, ionic strength, and the physicochemical properties of both the target molecule and endotoxin [2].Assay conditions also influence measured efficiency. Factors such as surfactants, chelators, and protein–LPS interactions can inhibit detection, leading to underestimation of endotoxin removal performance [4].
Strong anion exchange resins are commonly used for LPS removal due to their ability to bind negatively charged endotoxin while allowing neutral or positively charged proteins to pass through [2].

Figure 1: Factors influencing LPS removal efficiency
Affinity-based purification resins, including polymyxin B and histidine-functionalized media, provide highly specific endotoxin removal by directly interacting with lipid A [6]. Mixed-mode resins and hydrophobic interaction media may also contribute to endotoxin removal, particularly when LPS aggregates or associates with hydrophobic regions of proteins [3].
Evaluation of purification resins requires consistent experimental conditions, including identical feed material, assay methods, and reporting units such as EU/mL [2].Endotoxin removal is typically assessed by comparing pre- and post-purification levels, along with spike recovery controls to confirm assay validity and rule out masking effects [4]. Validated detection methods, such as recombinant Factor C assays, often operate within ranges as low as 0.005 EU/mL, enabling sensitive measurement of LPS removal performance [7].
LPS Removal vs Protein Recovery: Finding the Right Balance
A central challenge in LPS removal is balancing endotoxin removal with protein recovery. Competitive binding between endotoxin and target molecules can reduce yield if conditions favor product adsorption [6].
Optimization of buffer conditions is therefore necessary to maximize endotoxin removal while maintaining acceptable recovery levels. Key metrics for evaluating LPS removal include final endotoxin concentration (EU/mL), fold reduction, and binding capacity of the resin [2].
Additional considerations include flow rate sensitivity, reproducibility, and resistance to performance loss over repeated use, all of which impact overall endotoxin removal reliability [3].
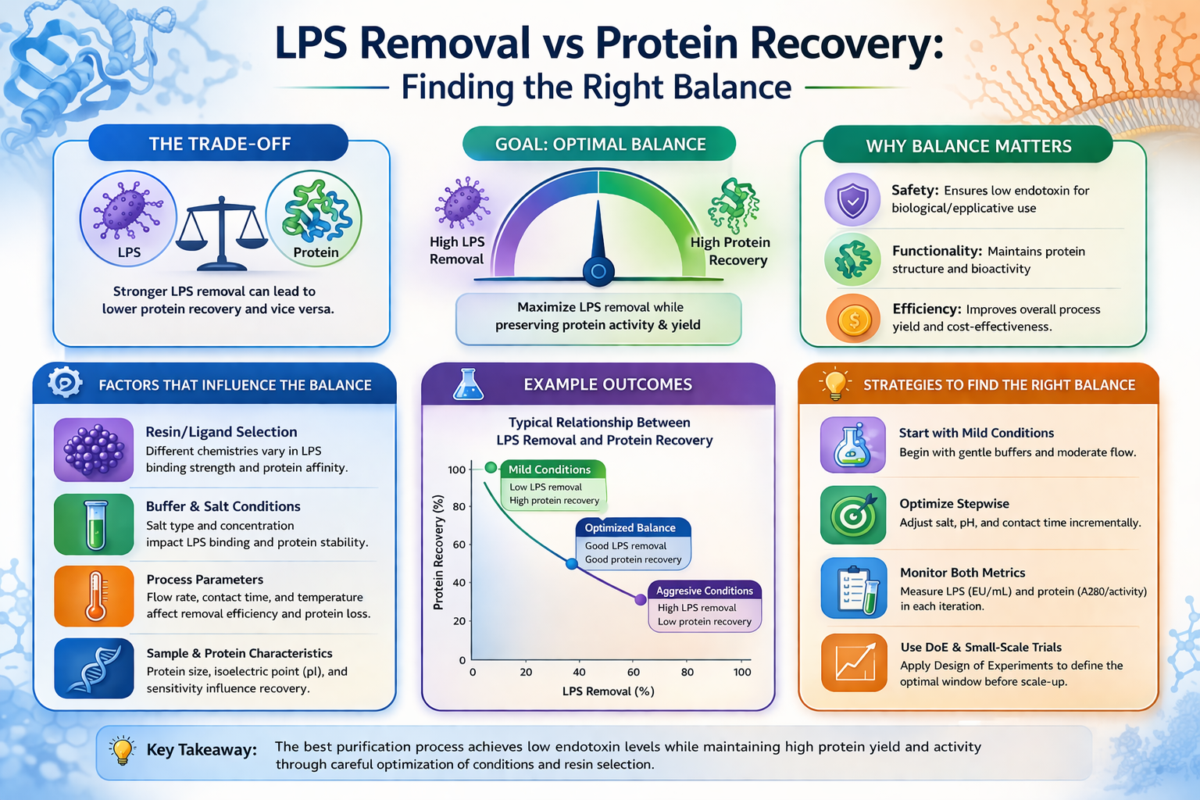
Figure 2: Balancing LPS removal and protein recovery
One of the most significant challenges in LPS removal is endotoxin masking, where assay interference leads to artificially low readings. This can result from buffer composition or LPS–protein interactions [4].
Another issue is variability in endotoxin contamination levels, which can complicate reproducibility and make it difficult to standardize endotoxin removal workflows.
Sample composition plays a critical role in LPS removal outcomes. Peptides and proteins with strong charge or hydrophobic properties may bind endotoxin, reducing removal efficiency [5]. In some cases, peptide–LPS interactions can alter assay detection, affecting measured endotoxin removal and complicating interpretation of results.
Best Practices for Improving LPS Removal Efficiency
Optimizing pH and ionic strength is one of the most effective strategies for improving LPS removal efficiency, as these parameters directly influence binding interactions [2]. Incorporating assay controls, such as spike recovery tests, ensures that endotoxin removal measurements reflect true performance rather than assay interference [4].
Combining multiple purification steps, including chromatography and affinity capture, can further enhance endotoxin removal in complex samples [3]. Selecting the appropriate resin for LPS removal depends on sample characteristics, required sensitivity, and downstream application requirements.
Anion exchange purification resins are often preferred for general endotoxin removal due to their high capacity and scalability [2]. However, affinity-based purification resins may be more effective for challenging samples with strong LPS binding, particularly when standard methods fail to achieve sufficient endotoxin removal [6].
Ultimately, the best approach to LPS removal involves empirical testing, as small changes in buffer conditions or sample composition can significantly alter endotoxin removal performance.
Frequently asked questions (FAQs) about LPS Removal
What is LPS removal and why is it important?
- LPS removal refers to the process of reducing or eliminating lipopolysaccharides (endotoxins) from peptide solutions or laboratory samples. This step is important because LPS, a component of Gram-negative bacterial cell walls, can trigger strong inflammatory responses and interfere with biological assays. Even trace levels of endotoxin can alter experimental outcomes, particularly in cell-based studies or immunological models, making effective LPS removal a critical part of quality control in peptide research workflows.
How do purification resins help with LPS removal?
- Purification resins are designed to selectively bind endotoxins through mechanisms such as hydrophobic interactions, ionic attraction, or affinity binding. Many LPS removal resins exploit the negatively charged and amphiphilic structure of lipid A, the toxic component of LPS, allowing the resin to capture endotoxin molecules while leaving peptides in solution. Common resin types include polymyxin B-based affinity resins, anion-exchange resins, and specialized endotoxin-binding matrices, each with different selectivity profiles and binding capacities.
What factors affect LPS removal efficiency?
- Several variables influence how effectively LPS can be removed from a sample. These include the initial endotoxin concentration, peptide composition and charge, buffer conditions (such as pH and ionic strength), and the presence of interfering substances like detergents or proteins. Additionally, endotoxin aggregation state and binding interactions with peptides can reduce accessibility, making removal more challenging. Proper optimization of conditions is often necessary to achieve consistent and reproducible results.
How should different purification resins be compared?
- Purification resins should be evaluated based on their endotoxin-binding capacity, specificity, recovery yield of the target peptide, and compatibility with the sample matrix. Key performance metrics include the percentage reduction in endotoxin levels (often measured in EU/mL), processing time, and potential for peptide loss or denaturation. Reproducibility and scalability are also important considerations, especially for workflows that require consistent batch-to-batch performance.
What should researchers consider when choosing a resin for LPS removal?
- When selecting a resin, researchers should consider the physicochemical properties of the peptide, including size, charge, and hydrophobicity, as well as the required endotoxin threshold for the intended application. Compatibility with solvents and buffers, ease of use, and regeneration capability are also relevant factors. In addition, researchers should evaluate whether the resin introduces any contaminants or interferes with downstream analytical methods, ensuring that LPS removal does not compromise overall sample integrity.
References
- Raetz CRH, Whitfield C. Lipopolysaccharide endotoxins. Annu Rev Biochem. 2002;71:635-700.
- Chen RH, Huang CJ, Newton BS, et al. Factors affecting endotoxin removal from recombinant therapeutic proteins by anion exchange chromatography. Protein Expr Purif. 2009;64(1):76-81.
- Ongkudon CM, Chew JH, Danquah MK. Chromatographic removal of endotoxins: A bioprocess engineer’s perspective. ISRN Chromatogr. 2012;2012:649746.
- Wespel M, Geiss M, Nägele M, et al. The impact of endotoxin masking on endotoxin removal. J Chromatogr A. 2022;1671:462995.
- Tamura H, Reich J, Nagaoka I. Bacterial endotoxin assays and peptide interactions. Juntendo Med J. 2016;62(2):132-140.
- Anspach FB, Hilbeck O. Removal of endotoxins by affinity sorbents. J Chromatogr A. 1995;711(1):81-92.
- Kang DH, Yun SY, Eum S, et al. Application of recombinant Factor C assay in endotoxin testing. Microorganisms. 2024;12(3):516.
