LPS Testing as Part of Peptide COA Documentation
Author: Dr. Numan S. Date: February 26, 2026

What Is COA Documentation?
Description: Understand how LPS testing fits into peptide COA documentation, what information COAs include, and why transparent documentation matters. A certificate of analysis is a lot-specific record that links a vial to a defined batch and a dated set of analytical results. In regulated settings, this documentation is meaningful only inside a system that controls suppliers, sampling, and change management.¹
The International Council for Harmonisation ICH Q7 GMP guide states that a supplier’s certificate of analysis can replace certain in-house tests only when suppliers are evaluated and the certificate’s reliability is periodically checked; it also specifies that the guide applies to active pharmaceutical ingredients for human drugs.¹ For many research purchases, COA paperwork is therefore evidence—not a guarantee—and it should be weighed against your assay’s sensitivity.
What Information Is Typically Included in Peptide COA Documentation
A peptide COA is strongest when it reports identity and impurity-related results in a reproducible way. European Medicines Agency guidance for synthetic peptide drug substances recommends confirming identity with at least two orthogonal methods and emphasizes suitability of impurity-control procedures.² This clarifies what “verified” ought to mean when you read research peptide documentation.²
Purity percentages are usually derived from chromatographic peak area, so method context matters. A key limitation is empirical: an R&D evaluation of synthetic peptides found that supplier purity claims on a certificate of analysis often diverged from in-house analyses, and some samples contained the wrong main compound.³ This is why peptide quality control often includes local confirmation of identity/purity for higher-stakes studies.³
What Is LPS and Why It Appears in COA Discussions
Lipopolysaccharide (LPS) is commonly discussed as bacterial endotoxin and is relevant because it can contaminate biomaterials through water, equipment, or microbial residues. U.S. Food and Drug Administration inspection guidance states that LPS is used synonymously with bacterial endotoxins and defines endotoxin activity in endotoxin units (EU).⁴ In COA language, lps detection is an activity measurement, not simply a mass specification.⁴
A Frontiers perspective explains why this matters experimentally: endotoxin can be thermoresistant, persist as an environmental contaminant, and bias experimental conclusions unless evaluated with appropriate assays and controls.⁵ For LPS‑sensitive biology, endotoxin testing is often required for interpretable data.⁵
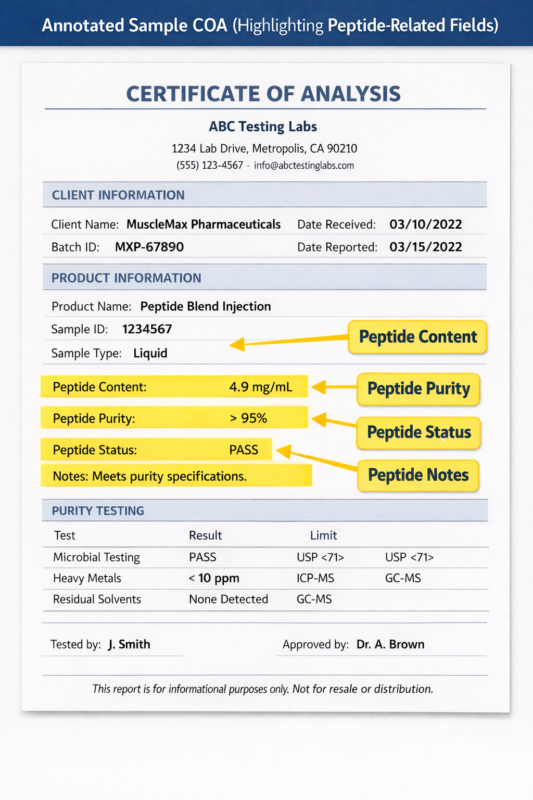
Figure 1: Sample Certificate of Analysis (COA)
How LPS Testing Fits Into Peptide COA Documentation
When LPS testing is performed, the result typically appears in COA Documentation as a single lps detection value (or “<” value) with units and a named method. That number is a point-in-time measurement on the tested sample; it cannot guarantee identical endotoxin activity after shipping or handling.⁵
A pragmatic rule is: if endotoxin testing is not listed, it has not been documented. This matters because the FDA’s endotoxin testing Q&A notes that storage and handling can affect detectability.⁶ In research peptide documentation, that becomes a decision point: accept uncertainty for low-sensitivity assays, or run/commission endotoxin testing when biology is likely to amplify trace contamination.⁶
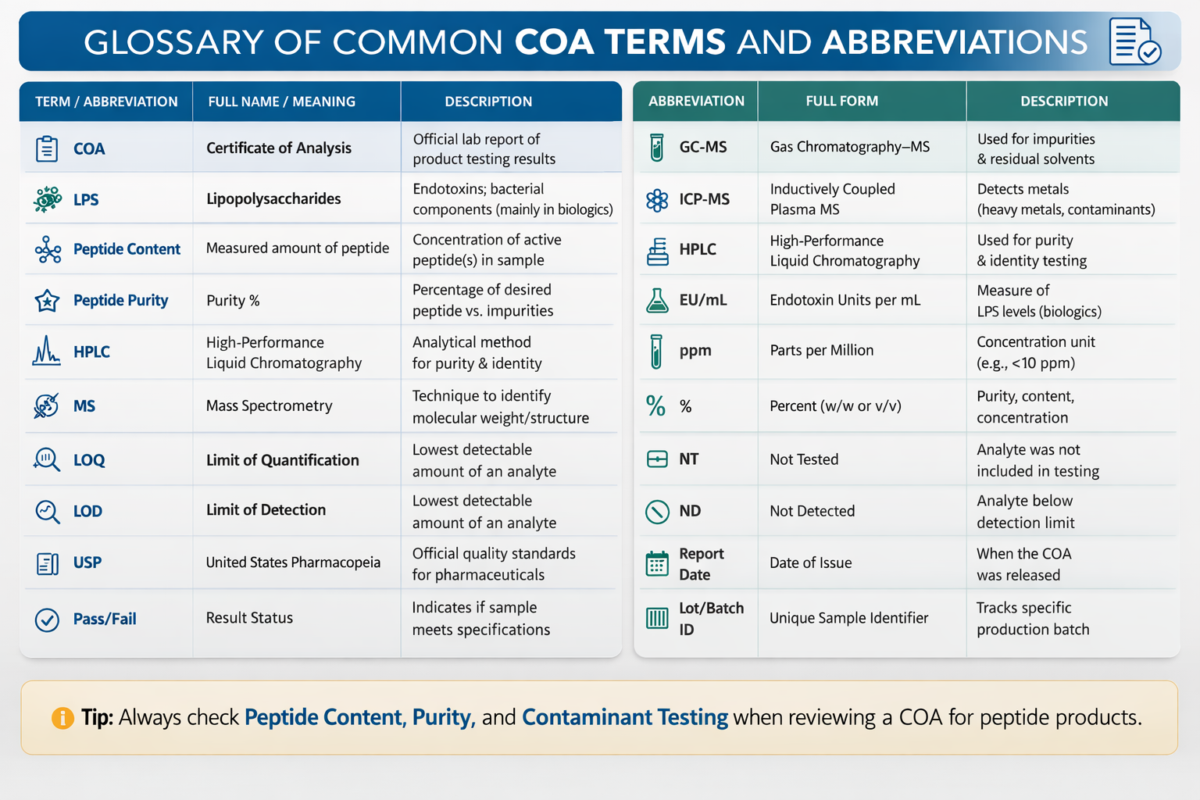
Figure 2: Glossary of Common Certificate of Analysis (COA) Terms & Abbreviations
Common LPS Testing Methods Referenced in COAs
Most reports that include lps detection reference Limulus amebocyte lysate (LAL)–based assays. The Frontiers perspective notes Factor C activation as the endotoxin-triggered step and describes common formats: gel‑clot, turbidimetric, and chromogenic readouts.⁵ Because formats differ in endpoint chemistry, assay choice/validation are part of laboratory testing standards for credible results.⁵
Recombinant Factor C assays are another route to lps detection, but matrix effects and validation determine comparability. A comparative study in complex human vaccine matrices reported recombinant Factor C assays overall at least equivalent to LAL yet showed that proteases/glucans can reduce suitability and require mitigation during validation.⁷
Interpreting LPS Results on a COA
Interpret lps detection by checking units, sample basis, and applied threshold. FDA inspection guidance defines EU and gives examples of limits expressed in EU/mL for certain pharmaceutical waters, underscoring that acceptable endotoxin levels depend on exposure context.⁴ For research, translate any EU/mg or EU/mL result into your actual dose and volume conditions before concluding “low risk.”⁴
Also assess whether the report supports laboratory testing standards: method name, assay format, units, and whether the result is quantified or below a limit. Because the FDA notes that handling affects detectability, and the literature emphasizes assay interference, consider confirmatory endotoxin screening when your system is LPS-sensitive or dosing is high.⁵,⁶ lps detection is most informative when paired with transparent method context.⁵
What COA Documentation Does Not Tell You
COA Documentation cannot guarantee representativeness of the aliquot you received, nor can it rule out post-analysis contamination. The synthetic-peptide evaluation provides an example: supplier documentation did not consistently match in-house results, supporting independent checks when data quality is critical.³ A certificate of analysis should therefore be treated as one piece of evidence, not as the full story.³
It also cannot tell you whether “small” endotoxin contamination is biologically irrelevant. In a controlled study, very low LPS concentrations (0.002–2 ng/mL) activated primary human CD1c+ dendritic cells, and the authors warned that even low endotoxin contamination may generate erroneous data in sensitive cells.⁸ This supports conservative lps detection for immune endpoints.⁸
Using COA Documentation to Support Better Research Decisions
For decisions, treat COA Documentation as one data point within research peptide documentation. A peptide COA with clear identity and purity may be sufficient for many assays; if the biology is LPS-sensitive, request (or generate) explicit endotoxin testing so lps detection is documented with units and a method name and can be compared across lots.⁵,⁶
Finally, anchor decisions in known confounders: identity/impurity issues (managed with peptide quality control) and inflammatory confounding from LPS (managed with targeted endotoxin testing). Evidence that very small LPS amounts can activate primary immune cells supports a conservative stance when interpreting endotoxin levels, especially at high dose or long exposure.⁸ A certificate of analysis is most valuable when it drives the right verification, not when it replaces it.⁸
Frequently asked questions (FAQs) about LPS Testing as Part of Peptide COA Documentation
What does a Certificate of Analysis (COA) show for peptide products?
- A Certificate of Analysis (COA) is a quality-control document that summarizes the analytical testing performed on a peptide batch. Typical COA fields include peptide identity confirmation (often by mass spectrometry), purity measurements from chromatographic analysis such as HPLC, peptide content or assay percentage, appearance, storage conditions, and batch or lot identification. Many COAs also include microbiological or endotoxin-related information depending on the manufacturer’s testing scope. For researchers, the COA provides transparency regarding the analytical methods used and confirms that the tested batch meets defined laboratory specifications.
What does LPS or endotoxin information on a COA represent?
- LPS results on a COA refer to testing for lipopolysaccharide endotoxin contamination. Endotoxin originates from the outer membrane of Gram-negative bacteria and can be introduced during peptide synthesis, purification, or handling processes. When reported on a COA, the endotoxin value typically indicates the measured endotoxin level expressed in endotoxin units (EU) per vial, milliliter, or milligram depending on the assay design. This information helps researchers evaluate potential biological interference when peptides are used in experimental models.
How is endotoxin typically measured for COA documentation?
- Most COA documentation referencing endotoxin testing relies on assays derived from the Limulus Amebocyte Lysate (LAL) reaction. These assays detect endotoxin through activation of a clotting cascade triggered by lipid A, the biologically active portion of LPS. Common formats include gel-clot, chromogenic, and turbidimetric assays. Some laboratories also reference recombinant Factor C (rFC) assays, which detect endotoxin through a synthetic form of the LPS-sensitive enzyme without using horseshoe crab lysate. These methods allow laboratories to quantify endotoxin levels with high sensitivity.
What information should researchers review when interpreting LPS results on a COA?
- When reviewing LPS data on a COA, researchers typically examine several factors: the reported endotoxin value, the assay method used, the detection limits of the assay, and the units of measurement. It is also useful to verify whether the result represents a single test or replicate measurements and whether the COA identifies the laboratory performing the analysis. Contextualizing these details helps researchers understand how endotoxin testing was conducted and how the results should be interpreted within experimental workflows.
Where does COA testing occur in the peptide lifecycle?
- COA documentation typically reflects testing conducted after peptide synthesis, purification, and lyophilization. Analytical testing is usually performed on the final peptide batch before distribution. The peptide lifecycle generally follows several stages: synthesis of the peptide sequence, purification (often by HPLC), lyophilization into a stable powder, analytical testing for identity and purity, and documentation of results in the COA. In this workflow, COA testing serves as the final analytical checkpoint that documents the characteristics of the batch being supplied for laboratory research.
What are common COA terms and abbreviations researchers may encounter?
- Several abbreviations frequently appear in peptide COA documentation. “HPLC” refers to high-performance liquid chromatography used to determine peptide purity. “MS” indicates mass spectrometry used to confirm molecular weight and identity. “EU” stands for endotoxin units used in LPS quantification. “LOD” refers to the limit of detection of an analytical assay. “RT” typically denotes retention time in chromatographic analysis. Understanding these terms helps researchers interpret COA data more effectively.
Why is transparent COA documentation important in peptide research?
- Transparent COA documentation allows researchers to verify analytical results associated with a peptide batch. Access to identity confirmation, purity measurements, and endotoxin testing data enables researchers to understand the analytical profile of the material used in experiments. Transparent documentation also allows comparisons between batches and helps maintain reproducibility in laboratory studies.
References
-
Al Musaimi O, Jaradat DMM. Advances in Therapeutic Peptides Separation and Purification. Separations. 2024;11(8):233. DOI: 10.3390/separations11080233
-
Guo C, Ling N, Tian H, et al. Comprehensive review of extraction, purification, structural characteristics, pharmacological activities, and application of seabuckthorn protein and peptides. Int J Biol Macromol. 2025;294:139447. DOI: 10.1016/j.ijbiomac.2024.139447
-
Barashkova AS, Rogozhin EA. Isolation of antimicrobial peptides from different plant sources: Does a general extraction method exist? Plant Methods. 2020;16(1):143. DOI: 10.1186/s13007-020-00687-1
-
Mant CT, Chen Y, Yan Z, et al. HPLC analysis and purification of peptides. Methods Mol Biol. 2007;386:3-55. DOI: 10.1007/978-1-59745-430-8_1
-
Isidro-Llobet A, Kenworthy MN, Mukherjee S, et al. Sustainability challenges in peptide synthesis and purification: from R&D to production. J Org Chem. 2019;84(8):4615-4628. DOI: 10.1021/acs.joc.8b03001
-
An J, Tsopmejio ISN, Wang Z, Li W. Review on extraction, modification, and synthesis of natural peptides and their beneficial effects on skin. Molecules. 2023;28(2):908. DOI: 10.3390/molecules28020908
