Pyrogen Testing vs Endotoxin Testing for Peptides
Author: Dr. Numan S. Date: January 29, 2026

What Is Endotoxin Testing?
Endotoxin testing is a more specific subset of pyrogen testing focused solely on bacterial endotoxins, especially lipopolysaccharides (LPS) from Gram-negative bacteria[7][8]. Endotoxins are among the most potent exogenous pyrogens; even minute amounts can trigger intense immune responses (fever, inflammation, septic shock)[9]. Endotoxin testing usually refers to the bacterial endotoxins test (BET) defined in pharmacopeial standards (USP <85>, EP 2.6.14, etc.), most often performed via the Limulus Amebocyte Lysate (LAL) assay[10]. The LAL test uses an extract of horseshoe crab blood cells that clots in the presence of LPS endotoxin, allowing detection of extremely low levels (sensitivities often 0.005 EU/mL or below)[10][11]. Endotoxin testing is critical in peptide quality control to ensure any product intended for injection has endotoxin levels below strict safety limits (e.g. 5 EU/kg for most injectable drugs)[12]. Unlike broad pyrogen tests, endotoxin assays target only LPS endotoxin. They are widely referenced in research and manufacturing because Gram-negative endotoxins are the most common pyrogenic contaminants and remain toxic even after sterilization[13]. In practice, endotoxin testing of peptides is an essential QC step to verify the absence of Gram-negative bacterial endotoxins in the final product.
What Is Pyrogen Testing?
Pyrogen testing refers to assays that detect any pyrogens – substances that induce fever when introduced into the body[1][2]. Pyrogens are broadly categorized as endogenous (originating within the body, e.g. cytokines) or exogenous (coming from outside, such as microbial contaminants)[3]. In the context of pharmaceuticals or research peptides, pyrogen testing usually means checking for fever-inducing contaminants in a sample before it’s administered to animals or humans. Historically, the official USP <151> Rabbit Pyrogen Test (RPT) was the first method: a sample is injected into rabbits and any rise in body temperature is measured[4]. This in vivo test can detect a wide range of pyrogens (not just bacteria) since rabbits respond with fever to many fever-causing substances. However, the rabbit test has drawbacks like low sensitivity, variability, and ethical concerns[5]. Modern regulations encourage in vitro alternatives such as the Monocyte Activation Test (MAT), which uses human blood cells to detect pyrogens by measuring cytokine release[6]. In summary, pyrogen testing encompasses methods (old and new) that ensure a peptide product will not cause febrile reactions by detecting any fever-inducing contaminants in the sample.
Pyrogen vs Endotoxin: What’s the Difference?
“Pyrogen” is an umbrella term for any substance that causes fever, whereas “endotoxin” refers specifically to the LPS molecule from Gram-negative bacteria – one particular type of pyrogen[14]. In other words, all endotoxins are pyrogens, but not all pyrogens are endotoxins[1]. For example, components from Gram-positive bacteria (like peptidoglycan or lipoteichoic acid), fungal cell wall glucans, and certain viral products can also act as pyrogens by inducing fever, even though they are non-endotoxin pyrogens. Endotoxins tend to be the most potent and clinically significant pyrogens; they are often considered the primary pyrogenic risk in pharmaceuticals[13].
Figure 1: Gram-negative (left) vs Gram-positive (right) bacteria under the microscope.
This is why so much emphasis is placed on endotoxin testing. However, focusing solely on LPS could miss other fever-causing contaminants. A key distinction is in testing: methods like RPT or MAT that detect “pyrogens” broadly will catch endotoxins and non-endotoxin pyrogens, whereas LAL is an endotoxin-specific test and will not detect pyrogens that are not LPS[15]. Practically, this means a peptide sample could pass an endotoxin (LAL) test but still contain other pyrogens capable of causing a reaction[1]. Understanding this difference is crucial: endotoxin testing is a narrow, targeted form of pyrogen testing. Endotoxin assays address the subset of pyrogens from Gram-negative bacteria, while pyrogen tests (in the broader sense) cover the full spectrum of fever-inducing impurities.
Why This Distinction Matters in Peptide Research
For scientists working with research peptides or developing peptide-based therapeutics, distinguishing between pyrogen testing and endotoxin testing is far from academic – it has real-world implications for safety and experimental validity. Many peptides are produced via synthetic methods under sterile conditions, yet they can harbor hidden endotoxins that standard purity assays won’t reveal[16][17]. A peptide might be >98% chemically pure and sterile (no live bacteria), and still cause a fever in a lab animal or trigger inflammatory cytokines in cell culture due to residual LPS endotoxin. In peptide quality control, researchers therefore routinely perform endotoxin testing on final peptide preparations to ensure that any observed biological effects are due to the peptide itself and not an LPS contaminant. The distinction also matters for interpreting results: an unexpected immune response in an experiment might prompt pyrogen testing (via MAT or RPT) if endotoxin-specific tests are negative. In summary, being mindful of pyrogens vs. endotoxins helps researchers avoid confounding variables. It ensures that “sterile” peptide preparations are also pyrogen-free, thereby preventing research artifacts and protecting animal models or patients from toxic inflammatory reactions[1]. This is why both types of tests are considered in peptide research and development – they address related but different risks that must be managed for reliable and safe results.
Common Methods Associated with Pyrogen Testing (High-Level Overview)
Several methods are used for comprehensive pyrogen testing. The classic method is the Rabbit Pyrogen Test (RPT), an in vivo assay. In the RPT, a sample of the peptide (or product) is injected intravenously into rabbits, and the animals’ body temperatures are monitored; a rise above a defined threshold indicates the presence of pyrogens[4]. Rabbits have a similar fever response threshold to humans, so this test can detect any pyrogen (endotoxin or otherwise) that causes fever. While robust in scope, the rabbit test is qualitative and relatively insensitive – it requires a sizable pyrogen load to trigger fever, and it cannot quantify how much pyrogen is present[5][18]. Moreover, due to animal welfare concerns and logistical downsides, the RPT has largely been supplanted by newer methods.
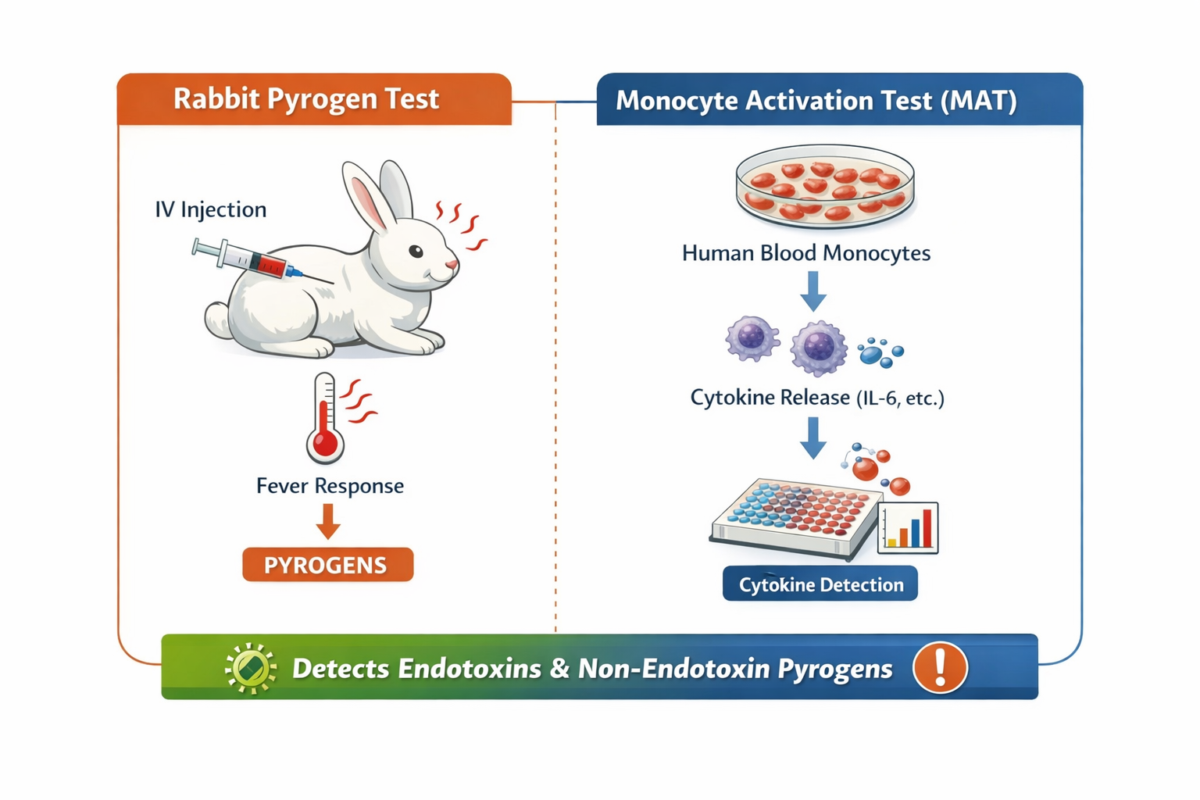
Figure 2: Pyrogen testing methods comparison.
The modern preferred method for broad pyrogen detection is the Monocyte Activation Test (MAT). The MAT is an in vitro assay that uses human immune cells (monocytes, either from fresh or cryopreserved blood) to simulate the human fever reaction. In the MAT, if pyrogens are present in the peptide sample, the monocytes will respond by releasing pro-inflammatory cytokines (such as IL-6), which can be measured (often by ELISA)[19][20]. The MAT detects both LPS endotoxins and non-endotoxin pyrogens with high sensitivity and quantitation. For example, the MAT will respond to Gram-positive bacterial fragments or fungal components that an LAL endotoxin test would miss[19]. Regulatory agencies recognize the MAT as an alternative to the rabbit test, and in fact the European Pharmacopoeia is actively moving toward phasing out the rabbit assay in favor of MAT for routine pyrogen testing[21]. In practice, labs performing pyrogen testing on peptide products may use the MAT for a broad assessment of pyrogenicity. Both RPT and MAT are considered “complete” pyrogen tests because their readouts (fever or cytokine release) cover the full range of exogenous pyrogens.
What Each Test Can and Cannot Detect
In summary, pyrogen tests like the Rabbit test and MAT can detect a wide range of fever-inducing agents (both endotoxins and non-endotoxin pyrogens), whereas endotoxin-specific tests (LAL or rFC) target only the LPS endotoxin from Gram-negative bacteria[15]. Each approach has its own detection scope. The RPT will catch anything that causes a fever in rabbits – for instance, it could flag a peptide sample contaminated with endotoxin, or one contaminated with say, a Gram-positive toxin, or even certain chemical contaminants. The MAT similarly will respond to any pyrogen that activates human monocytes (it has been shown to detect endotoxin as well as other microbial pyrogens like lipoteichoic acid from Staph.)[19]. By contrast, the LAL and rFC assays have blind spots: if a pyrogenic substance in the sample is not LPS, those tests yield a false sense of security. A clear consequence is that a “pass” in an endotoxin test does not guarantee the product is free of all pyrogens[1] – it only guarantees low levels of LPS endotoxin. Nonetheless, because endotoxins are typically the most potent pyrogens and the hardest to eliminate, many protocols focus on endotoxin testing as a proxy for pyrogen safety[13]. Best practice, however, is to use the appropriate test for the context: BET/LAL assays for routine monitoring of endotoxin, and a pyrogen test (MAT or RPT) when a broader safety evaluation is needed or when there’s suspicion that non-endotoxin pyrogens could be present[27].
Limitations and Considerations in Peptide Matrices
When applying these tests to peptide samples, researchers must consider the matrix effects and interference that peptides or their solvents might introduce. Peptides are often delivered in buffers or have additives (salts, residual solvents) that can potentially interfere with the LAL assay’s enzyme cascade. For example, a peptide solution with a yellow tint could confound a chromogenic LAL reading, or a turbid peptide suspension could interfere with a turbidimetric LAL measurement[28]. To address this, compendial methods require a validation step called “inhibition/enhancement” testing – essentially spiking known endotoxin into the peptide matrix to ensure the assay still recovers it accurately. Highly positively charged peptides (or formulations containing surfactants) can sometimes bind LPS and mask it from detection, yielding false-negative endotoxin results if not properly diluted. On the other hand, some peptide excipients might cause false positives in traditional LAL by triggering Factor G (for instance, glucans from cellulose filters – though this is avoided by using endotoxin-specific reagents or rFC)[26]. Another consideration is that very sensitive bioassays like the MAT might be affected if the peptide itself modulates monocyte activity (e.g. an immunomodulatory peptide could dampen or increase cytokine release independent of pyrogens). Researchers must factor in such possibilities when interpreting results. In peptide quality control, it is therefore crucial to choose the appropriate test and, if needed, dilute or treat the sample to fit the assay’s valid range[29]. By being mindful of these limitations, one can confidently use pyrogen and endotoxin tests to certify that a peptide product is safe and free of confounding contaminants.
Best Practices to Minimize Pyrogen and Endotoxin Risk
Controlling pyrogen and endotoxin contamination in peptide production requires a multi-pronged approach. Some best practices include:
- Sterile peptide practices: Maintain strict aseptic technique during synthesis, purification, and handling of peptides to prevent introducing bacteria. This includes working in cleanrooms, using sterile reagents and filters, and sanitizing equipment. However, because sterile practices alone don’t remove endotoxins, labs also ensure all containers, vials, and instruments are depyrogenated (for example, baking glassware at high heat or using certified pyrogen-free disposables)[30]. Adopting such sterile peptide practices from start to finish minimizes both microbial and endotoxin contamination risks.
- Use of Pyrogen-Free Materials: Wherever possible, use water-for-injection (WFI) or equivalent endotoxin-free water for preparing peptide solutions and buffers[37]. Water systems should be monitored and validated to produce water under the endotoxin limit. Similarly, use pyrogen-free vials, filters, and stoppers (many consumables come certified as “sterile and non-pyrogenic”). This prevents introduction of LPS from common sources like lab water or packaging materials.
- Routine Monitoring and Testing: Implement a peptide quality control plan that regularly tests for bioburden and endotoxins at critical steps. For instance, test the purification buffer and final peptide lot for endotoxins using LAL. If any test shows elevated endotoxin, investigate and re-clean equipment or halt the process. It’s also wise to perform MAT or rabbit tests on initial batches or when process changes occur[33], to catch any non-endotoxin pyrogens early. Consistent monitoring ensures that pyrogen levels stay in check throughout production.
- Equipment Cleaning and Sanitization: After each peptide synthesis or purification run, thoroughly clean chromatography columns, reactors, and lyophilizers with agents that remove endotoxins (such as dilute NaOH, which can hydrolyze LPS)[38]. Validate these cleaning procedures specifically for endotoxin removal. For example, HPLC columns used in peptide purification can accumulate endotoxin; flushing with high pH or chaotropic solutions can desorb and eliminate these contaminants[39]. Proper cleaning prevents pyrogen carry-over between batches.
By integrating these practices, peptide manufacturers and researchers can ensure their products remain both sterile and pyrogen-free. The goal is to minimize contamination at the source and have robust checks (through pyrogen and endotoxin testing) before any peptide product is deemed safe for biological use[40]. Taken together, such proactive measures greatly reduce the risk of endotoxin or other pyrogens derailing an experiment or harming a patient.
Common Misconceptions
- “If a product is sterile, it contains no pyrogens.” This is a dangerous misconception. Sterilization procedures kill live microbes but do not necessarily remove or destroy endotoxins and other pyrogens[30]. A peptide sample can pass sterility testing yet still cause a fever due to endotoxins. Hence, sterility alone is not a guarantee of safety – pyrogen testing (especially endotoxin assays) is still needed to confirm a pyrogen-free status.
- “Endotoxin testing isn’t important if a peptide is chemically pure.” Purity is not the same as safety. Even peptides synthesized to >99% purity can have high endotoxin levels from trace bacterial contamination during production[41][42]. These endotoxins are invisible to standard chemical analyses (HPLC, MS) used for purity. Thus, a peptide’s chemical purity does not rule out pyrogen contamination. Endotoxin testing is non-negotiable for any peptide intended for in vivo research or therapeutic use[17].
- “Endotoxin = pyrogen, so testing for endotoxins covers everything.” Not exactly. Endotoxins are indeed the most common pyrogens, but as discussed, other substances can cause fevers too. Relying solely on LAL tests could miss non-endotoxin pyrogens[15]. In critical applications (e.g. intrathecal drugs or novel biologics), a broader pyrogen test (MAT or RPT) may be required to ensure absolutely no fever-causing agents are present[27].
Frequently asked questions (FAQs) about Pyrogen Testing vs Endotoxin Testing for Peptides
What is pyrogen testing and how is it different from endotoxin testing?
- Pyrogen testing evaluates whether a sample can trigger a fever-inducing (pyrogenic) response, regardless of the chemical source. It is designed to detect a broad range of fever-causing substances, including bacterial components and certain non-bacterial contaminants. Endotoxin testing, by contrast, specifically detects lipopolysaccharide (LPS) from Gram-negative bacteria. In short, endotoxin testing targets one well-defined pyrogen, while pyrogen testing assesses a wider spectrum of fever-causing agents.
Are all endotoxins considered pyrogens?
- Yes. Endotoxins are a subset of pyrogens because LPS reliably induces strong inflammatory and febrile responses in biological systems. However, not all pyrogens are endotoxins. Other substances—such as components from Gram-positive bacteria, fungal cell wall fragments, or certain chemical impurities—can also act as pyrogens without being LPS.
Why might a sample pass endotoxin testing but still contain pyrogens?
- A sample may pass endotoxin testing if it contains little or no LPS, yet still harbor non-endotoxin pyrogens. These can arise from microbial remnants, raw material impurities, or processing-related contaminants that are invisible to endotoxin-specific assays. In such cases, endotoxin results appear acceptable even though the sample still has the potential to cause a pyrogenic response.
What are common sources of pyrogen contamination in peptide research?
- Pyrogens can be introduced at multiple stages, including during peptide synthesis, purification, or handling. Common sources include contaminated water or buffers, improperly cleaned glassware, raw materials with residual microbial components, and prolonged exposure of solutions to non-sterile environments. Even dead bacteria or degraded cell wall fragments can contribute pyrogenic material.
What practices help reduce pyrogen and endotoxin risk in the lab?
- Risk is reduced by using high-purity, pyrogen-free reagents and water; maintaining strict cleaning and depyrogenation protocols for glassware; minimizing open handling steps; and working in controlled, clean environments. Routine quality control testing, combined with careful storage and documentation of materials, further helps limit both endotoxin and broader pyrogen exposure in peptide research workflows.
References
- BradyKnows Medical. What is the difference between pyrogens and bacterial endotoxins in medical devices? (2023) – Explains that endotoxins are a subset of pyrogens and that sterilization does not remove all pyrogenic substances.
- Fujifilm Wako Chemicals USA. Understanding the difference between sterility, bioburden, pyrogen, and bacterial endotoxin testing (Blog, 2025) – Notes that rabbit pyrogen tests detect both endotoxin and non-endotoxin pyrogens, whereas LAL is specific to endotoxin.
- Nelson Labs. What are the common test methods for pyrogen testing and how to choose between them? (Blog, 2023) – Emphasizes that all pyrogen tests will detect endotoxins, but endotoxin-specific tests won’t detect other pyrogens; also that endotoxins remain toxic after sterilization.
- Peptide Partners (Substack). The Hidden Danger in High-Purity Peptides: Why Endotoxin Testing Is Non-Negotiable (2025) – Describes how even 95–99% pure research peptides can harbor endotoxins causing severe inflammatory responses, and stresses the importance of USP <85> endotoxin testing.
- R&D World. Controlling Endotoxin Contamination During Peptide Manufacturing (2012) – Discusses sources of endotoxin in peptide production and recommends using pyrogen-free materials and monitoring water and equipment to minimize endotoxin risk.
- Solati, S. et al. Innate Immun. 28(3-4):130–137 (2022) – Research demonstrating the Monocyte Activation Test detects both endotoxins and non-endotoxin pyrogens and can reveal synergistic pyrogenic effects undetected by LAL alone.
- Pacific BioLabs. LAL and Bacterial Endotoxins Testing – Pharm/Biopharm (Web) – Defines endotoxins as LPS from Gram-negative bacteria and notes water as a source of pyrogens, highlighting the need for routine endotoxin testing of water systems.
- Fujifilm Wako Chemicals USA. Comparing the LAL Method with the Pyrogen Test on Rabbits (Blog) – Observes that LAL is more sensitive than the rabbit pyrogen test and explains the historical shift toward LAL for endotoxin detection in parenteral drugs.
- FDA Guidance for Industry. Pyrogen and Endotoxins Testing: Questions and Answers (2012) – Recommends risk-based use of MAT or RPT if non-endotoxin pyrogens may be present, and otherwise reliance on endotoxin-specific testing for routine batch release.
- BMG Labtech. Pyrogens and Pyrogen Testing (Blog, 2025) – Provides an overview of pyrogen types and testing methods, including a figure contrasting animal vs. in vitro tests and endotoxin-specific vs. broad pyrogen detection.
