Two-Stage LPS Filtration for Ultra-Low Endotoxin Peptides
Author: Dr. Numan S. Date: April 9, 2026

What Is LPS Filtration?
Lipopolysaccharide (LPS) filtration is a targeted separation technique used to remove endotoxins—large amphiphilic molecules derived from Gram-negative bacterial outer membranes—from aqueous peptide solutions. LPS molecules form aggregates and micelles that can range from tens to hundreds of nanometers, making them challenging contaminants during peptide purification [1]. Effective LPS filtration leverages size exclusion, charge interactions, and adsorption mechanisms to achieve endotoxin reduction without compromising peptide integrity.
Why LPS Filtration Matters for Ultra-Low Endotoxin Peptides
Maintaining ultra-low endotoxin levels is critical for analytical reproducibility and downstream biological testing. Even trace levels of LPS can activate immune signaling pathways, skewing in vitro assay outcomes and confounding interpretation [2]. Incorporating LPS filtration into peptide purification workflows ensures that contaminants are removed at stages where endotoxin burden is highest, supporting consistent experimental conditions and minimizing assay variability.
How Two-Stage LPS Filtration Works
Two-stage LPS filtration involves sequential processing through complementary filter systems designed to remove endotoxin species with different physicochemical properties. The first stage typically targets large aggregates using ultrafiltration or membrane-based exclusion, while the second stage employs affinity or adsorption filters that bind residual endotoxin molecules [3]. This layered approach enhances overall endotoxin reduction compared to single-pass filtration methods.
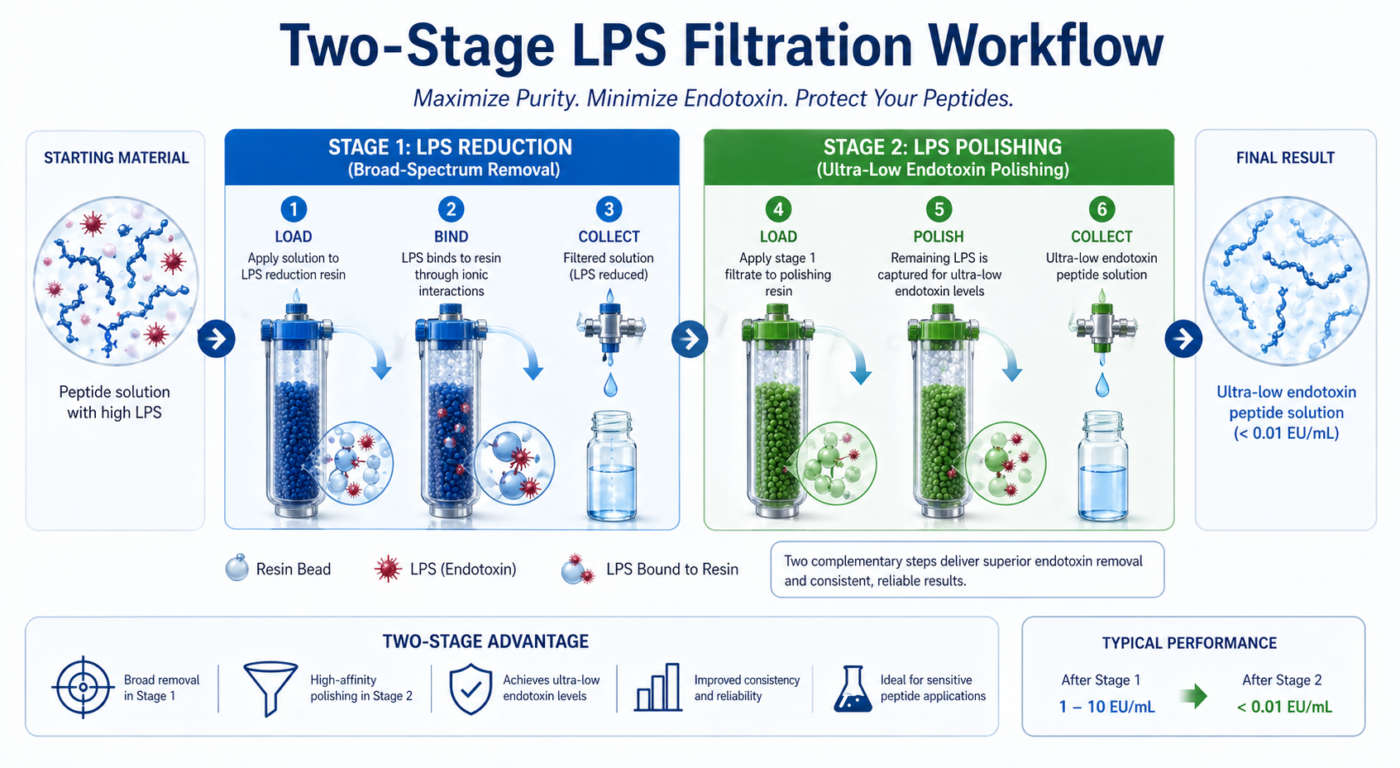
Figure 1: Workflow diagram showing each stage of a two-stage LPS filtration process.
By combining mechanisms such as pore-size exclusion and electrostatic binding, two-stage LPS filtration provides broader coverage of endotoxin species. This strategy is particularly effective when dealing with heterogeneous LPS populations that vary in size, charge, and aggregation state.
A two-stage LPS filtration strategy differs from conventional single-step approaches by addressing the limitations inherent in any one filtration mechanism. Size-based filtration alone may allow smaller endotoxin fragments to pass, while affinity filters can become saturated when exposed to high LPS loads [4]. Sequential integration of both methods distributes the removal burden, improving efficiency and extending filter lifespan.
Additionally, the staged design allows optimization at each step, including flow rate, buffer composition, and pressure conditions. This modularity enables researchers to tailor the filtration workflow to specific peptide characteristics, enhancing reproducibility and scalability in peptide purification processes.
How LPS Filtration Supports Peptide Purity Goals
LPS filtration plays a direct role in achieving stringent peptide purity standards by removing biologically active contaminants that are not detectable by conventional chromatographic purity metrics. High-performance liquid chromatography (HPLC) confirms chemical purity but does not quantify endotoxins, necessitating orthogonal approaches for endotoxin reduction [5].

Figure 2: Table mapping common peptide workflow challenges to filtration strategy choices
By integrating LPS filtration into purification pipelines, researchers can ensure that both chemical and biological purity criteria are met. This is particularly important for peptides intended for sensitive cellular assays, where even low endotoxin concentrations can alter gene expression profiles and cytokine production.
Common filter types used in LPS filtration workflows include ultrafiltration membranes, depth filters, and affinity resins functionalized with polymyxin B or other endotoxin-binding ligands. Ultrafiltration membranes operate based on molecular weight cutoffs, removing large LPS aggregates while allowing peptides to pass [6].
Affinity filters, in contrast, exploit the strong interaction between LPS lipid A regions and cationic ligands, enabling selective binding and removal. Combining these filter types within a two-stage LPS filtration system maximizes endotoxin reduction across diverse sample conditions.
Why One Filtration Step May Not Be Enough
Single-stage filtration often fails to achieve comprehensive endotoxin reduction due to the structural diversity of LPS. Aggregated forms may be retained effectively, but monomeric or fragmented endotoxins can bypass size-based filters [7]. Conversely, affinity filters may exhibit reduced performance when overloaded or exposed to complex matrices.
Two-stage LPS filtration mitigates these limitations by ensuring that different endotoxin populations are addressed sequentially. This redundancy is essential for achieving ultra-low endotoxin thresholds required in high-sensitivity research applications.
The efficiency of LPS filtration is strongly influenced by sample properties such as pH, ionic strength, peptide concentration, and the presence of organic solvents. LPS aggregation behavior is highly dependent on buffer composition, which can alter filter performance [8].
For example, high salt concentrations may disrupt electrostatic interactions between LPS and affinity ligands, reducing binding efficiency. Similarly, high peptide concentrations can compete for adsorption sites or increase solution viscosity, affecting flow dynamics within the filtration workflow.
What To Know About LPS Filtration Efficiency and Recovery
Balancing endotoxin reduction with peptide recovery is a central consideration in LPS filtration. Excessive binding or retention can lead to product loss, particularly for hydrophobic peptides that may interact with filter materials [9]. Optimization of operating conditions—including pressure, flow rate, and buffer composition—is essential to maintain high recovery yields.
Efficiency is typically evaluated by measuring endotoxin levels before and after filtration using kinetic chromogenic assays, alongside peptide concentration analysis. These dual metrics provide a comprehensive assessment of filtration performance.
Despite its advantages, two-stage LPS filtration presents challenges such as filter fouling, reduced flow rates, and variability in endotoxin binding capacity. Fouling can occur due to protein or peptide accumulation on membrane surfaces, leading to decreased permeability and inconsistent performance.
Another challenge is the potential for endotoxin desorption under changing buffer conditions, which can reintroduce contaminants downstream. Careful control of process parameters and validation of each filtration step are necessary to ensure reliable endotoxin reduction.
Best Practices for Building a Reliable LPS Filtration Workflow
A robust filtration workflow should incorporate pre-filtration steps to remove particulates, followed by staged LPS filtration with validated filters. Monitoring key parameters such as pressure differentials, flow rates, and endotoxin levels at each stage enhances process control.
Standardizing buffer systems and maintaining consistent operating conditions are also critical for reproducibility. Incorporating in-process testing allows early detection of deviations, enabling corrective actions before final product analysis.
Two-stage LPS filtration is most beneficial in applications requiring ultra-low endotoxin levels, such as cell-based assays, immunological studies, and analytical validation workflows. It is particularly valuable when peptides are produced in environments with high endotoxin exposure or when downstream applications are highly sensitive to contamination.
In these contexts, integrating two-stage LPS filtration into peptide purification pipelines provides a reliable strategy for achieving stringent endotoxin specifications while preserving peptide integrity and functionality.
Frequently asked questions (FAQs) about Two-Stage LPS Filtration for Ultra-Low Endotoxin Peptides
What is LPS filtration?
- LPS filtration refers to the removal of lipopolysaccharides (endotoxins) from peptide solutions using specialized purification methods. Because LPS molecules are large, amphiphilic, and can form aggregates, they are difficult to eliminate with standard sterile filtration alone. LPS filtration typically involves affinity resins, charged membranes, or adsorption-based materials designed to selectively bind and remove endotoxins while preserving the peptide of interest.
How does two-stage LPS filtration work for peptides?
- Two-stage LPS filtration uses a sequential approach to maximize endotoxin removal. In the first stage, a high-capacity removal step (such as an endotoxin-binding resin or charged depth filter) reduces the bulk LPS load. In the second stage, a polishing step—often a finer membrane or high-specificity affinity material—removes residual endotoxin that escapes the first pass. This layered strategy improves overall clearance while maintaining tighter control over peptide purity and consistency.
Why would a peptide workflow use two filtration stages instead of one?
- A single filtration step may not achieve ultra-low endotoxin thresholds, especially when starting LPS levels are high or when peptides interact with endotoxins. Two-stage filtration provides redundancy and improved efficiency by addressing both high-load removal and low-level polishing. It also reduces the burden on any one material, which can help prevent saturation, breakthrough, or inconsistent results in sensitive analytical or research workflows.
When does two-stage LPS filtration make sense for ultra-low endotoxin peptide goals?
- Two-stage filtration is most appropriate when workflows require very low endotoxin levels (e.g., sub-EU/mL targets), when initial contamination is variable or elevated, or when downstream applications are highly sensitive to endotoxin presence. It is also beneficial for complex peptides that may shield or bind LPS, making single-step removal less reliable. In these cases, combining a bulk reduction step with a polishing stage helps ensure reproducible, high-purity outcomes aligned with stringent research standards.
References
- Petsch D, Anspach FB. Endotoxin removal from protein solutions. J Biotechnol. 2000;76(2-3):97-119.
- Rietschel ET, Brade H. Bacterial endotoxins. Sci Am. 1992;267(2):54-61.
- Magalhães PO, Lopes AM, Mazzola PG, et al. Methods of endotoxin removal from biological preparations. J Pharm Pharm Sci. 2007;10(3):388-404.
- Liu S, Tobias R, McClure S, et al. Removal of endotoxin from recombinant protein preparations. Clin Biochem. 1997;30(6):455-463.
- Swartz ME. HPLC methods for peptide analysis. J Chromatogr B. 2005;814(1):1-15.
- van Reis R, Zydney A. Bioprocess membrane technology. J Membr Sci. 2007;297(1-2):16-50.
- Gorbet MB, Sefton MV. Endotoxin: the uninvited guest. Biomaterials. 2005;26(34):6811-6817.
- Petsch D, Deckwer WD, Anspach FB. Protein adsorption on endotoxin removal membranes. J Biotechnol. 1998;64(2-3):125-139.
- Karunaratne DN, et al. Adsorptive losses during endotoxin removal processes. Biotechnol Bioeng. 1999;63(2):222-230.
