β-Glucan Interference: Why rFC Testing May Be Superior
Author: Dr. Numan S. Date: April 30, 2026

Endotoxin contamination remains a major analytical concern during pharmaceutical and peptide production. Reliable detection methods are essential because bacterial endotoxins can affect product quality, process validation, and laboratory consistency [1,2]. Traditional Limulus amebocyte lysate assays have been widely used for decades, but some methods may react to non-endotoxin substances such as β-glucans [3].
![Scientific comparison of rFC and traditional LAL endotoxin assays in a peptide laboratory environment]
What Is rFC Testing and β-Glucan Interference in Endotoxin Testing?
rFC testing is an endotoxin detection method based on recombinant factor C technology rather than horseshoe crab blood-derived lysate. The assay uses a recombinant version of factor C, the endotoxin-sensitive protein naturally involved in the Limulus clotting cascade [4].
Because rFC testing targets endotoxin activation directly, the method avoids some non-specific pathways associated with traditional lysate systems. Recombinant factor C assays are increasingly used in pharmaceutical quality control and research laboratories due to their specificity and consistency [4,5].
β-glucan interference occurs when glucan-containing substances activate portions of the LAL cascade unrelated to bacterial endotoxins. Certain traditional assays contain factor G, which reacts to β-glucans from fungal or environmental sources [3].
This unintended activation can complicate endotoxin testing because signals may appear elevated even when endotoxin levels remain low. β-glucan interference has been documented in multiple pharmaceutical and biological matrices [3,6].
Why β-Glucan Interference Can Complicate Peptide Testing
Some peptide ingredients, excipients, filters, and laboratory materials may introduce glucan-containing contaminants during manufacturing or handling [6]. In complex analytical environments, these materials can affect assay interpretation and increase the risk of false-positive endotoxin signals.
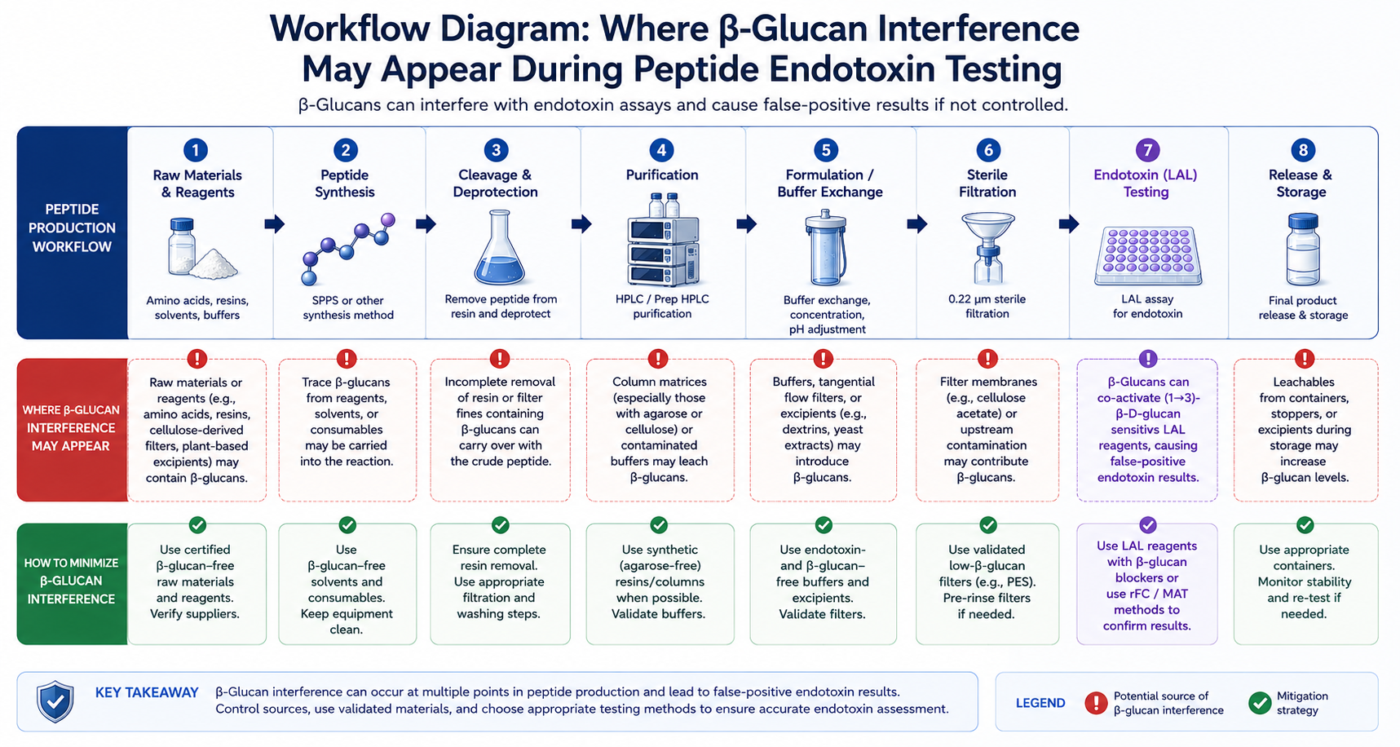
Figure 1: Workflow diagram showing where β-glucan interference may appear during peptide endotoxin testing.
For peptide workflows involving highly purified materials or sensitive analytical thresholds, distinguishing endotoxin activity from glucan reactivity becomes important. Excess assay noise may complicate validation and batch comparison studies [2,6].
Traditional LAL assays rely on a cascade containing both factor C and factor G pathways. While endotoxins activate factor C, β-glucans can activate factor G [3]. In contrast, rFC testing isolates only the endotoxin-sensitive mechanism. Because recombinant factor C lacks factor G-related reactivity, the assay may provide more selective endotoxin detection in certain applications [4,5].
How To Evaluate rFC Testing vs LAL for Peptide Applications
Method evaluation should include sensitivity, interference testing, matrix compatibility, and recovery performance. Comparative studies often assess equivalence between rFC testing and compendial lysate methods across multiple sample types [5,7]. Laboratories may also evaluate assay reproducibility, reagent stability, and detection thresholds before implementation. Validation protocols remain essential regardless of the selected platform [2].
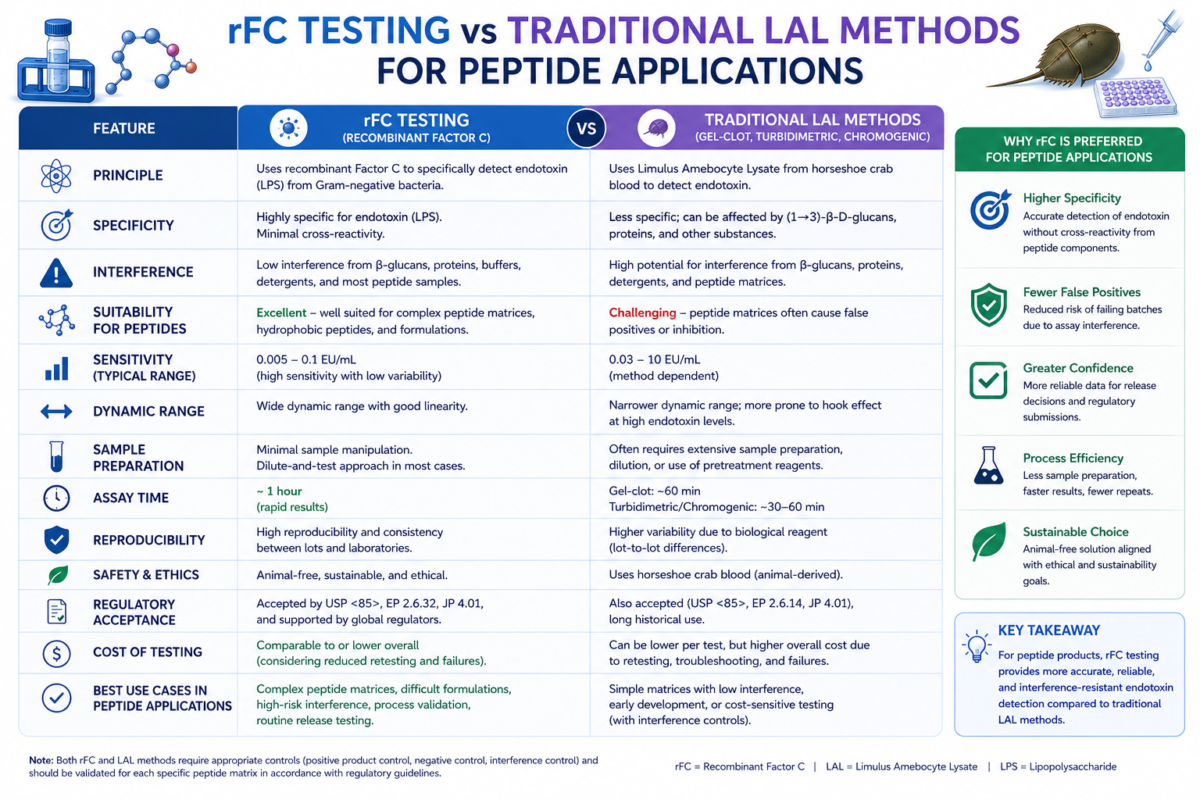
Figure 2: Comparison chart of rFC testing vs traditional LAL methods for peptide applications.
Before adopting alternative methods, teams should evaluate regulatory guidance, instrument compatibility, and internal validation requirements. Demonstrating equivalence to established endotoxin testing procedures is typically necessary [2,8]. Spike recovery studies, inhibition-enhancement testing, and matrix suitability assessments are commonly used during validation. These studies help confirm that sample components do not interfere with endotoxin detection accuracy [8].
What Types of Peptide Workflows May Benefit From rFC Testing?
Peptide workflows involving fermentation-derived ingredients, biologically active compounds, or complex purification systems may benefit from increased assay specificity. rFC testing may also support laboratories that routinely encounter glucan-containing filters or media components [5].
Research facilities emphasizing highly sensitive analytical thresholds sometimes evaluate recombinant factor C systems to reduce non-specific assay activation. This approach can simplify troubleshooting during quality investigations [7].
Factor G is sensitive to polysaccharide structures commonly associated with fungal cell walls and environmental glucans [3]. When activated, factor G can trigger downstream reactions that resemble endotoxin activity even though bacterial lipopolysaccharides are absent.
This mechanism explains why β-glucan interference remains an important consideration during endotoxin testing. Laboratories working with biologically derived compounds often evaluate glucan exposure risks when selecting assay platforms [6].
Why rFC Testing May Be More Specific for Endotoxin Detection
Studies comparing rFC testing with conventional lysate assays have demonstrated strong correlation for endotoxin recovery while reducing glucan-associated responses [5,7]. This specificity can improve analytical confidence in environments where glucan contamination is possible.
Another advantage is assay consistency. Since recombinant factor C is manufactured through controlled recombinant production systems, batch variability may be reduced compared with naturally sourced lysate materials [4].
Peptide workflows involving fermentation-derived ingredients, biologically active compounds, or complex purification systems may benefit from increased assay specificity. rFC testing may also support laboratories that routinely encounter glucan-containing filters or media components [5].
Research facilities emphasizing highly sensitive analytical thresholds sometimes evaluate recombinant factor C systems to reduce non-specific assay activation. This approach can simplify troubleshooting during quality investigations [7].
Common Challenges in Endotoxin Testing for Peptides
Peptide samples can present analytical difficulties because formulation ingredients, buffers, and purification residues may affect assay performance. Sample dilution requirements may also alter detectable endotoxin concentrations [6].
Environmental contamination control remains another major challenge. Water systems, containers, and filtration materials may contribute background interference during endotoxin testing if strict controls are not maintained [1].
rFC testing may be advantageous when glucan-related reactivity complicates traditional assay interpretation. Laboratories seeking improved specificity may prefer recombinant systems in applications where β-glucan interference is a recurring concern [5,7].
Facilities focused on sustainability have also shown interest in recombinant factor C technologies because they reduce reliance on horseshoe crab-derived lysate materials [4].
Teams considering rFC testing should understand that successful implementation depends on proper validation, analyst training, and matrix compatibility studies. The method should be evaluated within the context of the intended analytical application [2,8].
Although traditional assays remain widely used, recombinant approaches continue gaining acceptance across pharmaceutical and biotechnology environments. Careful evaluation of assay specificity, interference risk, and workflow requirements can help determine whether rFC testing is appropriate for a given laboratory setting [5].
Frequently asked questions (FAQs) about Why rFC Testing May Be Superior
What is rFC testing?
- Recombinant Factor C (rFC) testing is an endotoxin detection method that uses a genetically engineered version of Factor C, the endotoxin-sensitive component originally found in horseshoe crab blood. When bacterial endotoxins are present, recombinant Factor C becomes activated and generates a measurable fluorescent signal. rFC testing is commonly used to evaluate endotoxin contamination in pharmaceutical, biotechnology, and peptide research workflows because it can provide sensitive and specific endotoxin detection without relying on harvested horseshoe crab lysate.
What is β-glucan interference in endotoxin testing?
- β-glucan interference occurs when certain endotoxin assays react to β-glucans instead of lipopolysaccharides (LPS). β-glucans are polysaccharides commonly derived from fungi, cellulose filters, laboratory materials, or environmental contamination. In some traditional Limulus Amebocyte Lysate (LAL) assays, β-glucans may activate Factor G pathways and generate false-positive endotoxin signals. This interference can complicate data interpretation, particularly in peptide workflows where excipients, buffers, or filtration materials may introduce glucan contamination.
Why may rFC testing be superior for some peptide workflows?
- rFC testing may offer advantages for peptide workflows because it specifically targets endotoxin activation through recombinant Factor C without involving the Factor G pathway associated with β-glucan sensitivity. This increased specificity may help reduce the risk of false-positive results caused by glucan contamination. In peptide manufacturing or analytical environments where highly sensitive endotoxin monitoring is required, rFC testing may support clearer interpretation of endotoxin data and improve consistency across quality control workflows.
How does rFC testing differ from traditional LAL methods?
- Traditional LAL methods rely on lysate extracted from horseshoe crab blood and can involve multiple biological activation pathways, including both endotoxin-sensitive and β-glucan-sensitive mechanisms. In contrast, rFC testing uses only recombinant Factor C, which is designed to respond specifically to endotoxins. Because rFC assays remove the Factor G pathway associated with glucan activation, they may provide improved specificity in certain applications. rFC methods may also offer advantages related to reagent consistency, sustainability, and reduced biological variability between assay lots.
What should teams consider before adopting rFC testing for peptides?
- Before implementing rFC testing, teams should evaluate assay sensitivity requirements, peptide matrix compatibility, regulatory expectations, and method validation needs. Some peptide formulations or buffers may still interfere with fluorescence-based detection systems, making validation studies important before full adoption. Laboratories should also compare rFC performance against existing endotoxin testing workflows to confirm accuracy, reproducibility, and recovery performance under real operating conditions. Careful method qualification can help determine whether rFC testing is appropriate for a specific peptide quality control strategy.
References
- Magalhães PO, Lopes AM, Mazzola PG, Rangel-Yagui C, Penna TC, Pessoa A Jr. Methods of endotoxin removal from biological preparations: a review. J Pharm Pharm Sci. 2007;10(3):388-404. Available from: PubMed Central
- United States Pharmacopeia (USP) <85> Bacterial Endotoxins Test
- Bolden J, Smith K. Mechanisms of factor G activation and β-glucan interference in Limulus amebocyte lysate assays. Biotechnol Appl Biochem. 2017;64(3):321-329.
- Maloney T, Novitsky TJ. Recombinant factor C endotoxin testing: historical perspective and comparative analysis. PDA J Pharm Sci Technol. 2016;70(2):156-165. Available from: PubMed
- Chen J, Vinther A, Sørensen UB. Comparison of recombinant factor C and Limulus amebocyte lysate assays for endotoxin detection in pharmaceutical samples. Biologicals. 2021;70:1-7. Available from: ScienceDirect
- Williams KL. Endotoxin Detection and Control in Pharma, Limulus, and Mammalian Systems. Springer; 2019. Available from: Springer Book Listing
- Dubczak J, Kooman J, Steinschulte A. Evaluation of recombinant factor C assays for low-interference endotoxin detection. Pharm Technol. 2020;44(7):28-34. Available from: Pharmaceutical Technology
- European Pharmacopoeia Commission – Bacterial Endotoxins 2.6.14
